Apixaban Recalled Following Complaints of Dose-Packaging Mismatch
A single lot of 5-mg tablets is being recalled across the US after a complaint about a pill bottle containing the wrong strength.
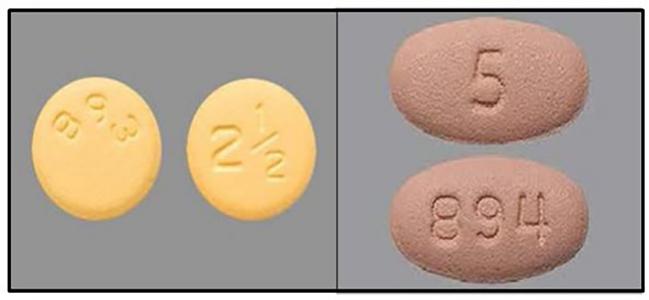
A single lot of apixaban 5-mg tablets is being recalled nationwide following a customer complaint about the wrong dosage, the manufacturer has announced.
The US Food and Drug Administration (FDA) issued a Safety Alert warning physicians and consumers about the recall. According to a statement from manufacturer Bristol-Myers Squibb, the voluntary recall is a “precautionary” measure after a customer complained that an apixaban (Eliquis) bottle labeled as containing the 5-mg dosage actually contained 2.5-mg tablets.
The two dosages have “distinct visible differences,” the company notes, including color, pill size, and markings—the lower dose is yellow and round in shape and has the numbers “893” on one side and “2½” on the other. By contrast, the 5-mg pill is stamped “894” with “5” on the reverse, is pink in color, and has an oval shape.
Both pills are “biconvex,” meaning that they’re thicker in the middle than at their edges.
“Patients who are prescribed Eliquis 5 mg for [atrial fibrillation] and take an Eliquis 2.5 mg tablet instead, particularly for a prolonged period, would have an increased probability of stroke, a moving blood clot, or death,” the company states. And for patients with deep vein thrombosis and pulmonary embolism, they say, “underdosing of the drug could lead to an increased risk of a growing or moving blood clot. Should that occur, it could be life-threatening or reversible depending on the severity and location of the blood clot.”
To date, the company adds, there have not been any reports of injuries or illnesses related to this issue, and pharmacies and wholesalers have been contacted with instructions for returning product.
Photo Credit: Bristol-Myers Squibb
Shelley Wood was the Editor-in-Chief of TCTMD and the Editorial Director at the Cardiovascular Research Foundation (CRF) from October 2015…
Read Full BioSources
US Food and Drug Administration. Bristol-Myers Squibb voluntarily recalls one lot of Eliquis (apixaban) 5 mg tablets. Published on: June 10, 2017. Accessed on: June 13, 2017.


Comments