CANTOS: Big Drop in CVD Events Among Canakinumab Responders
The analysis identified “biologic responders” to therapy, suggesting a single dose might be used to determine who would benefit from treatment.
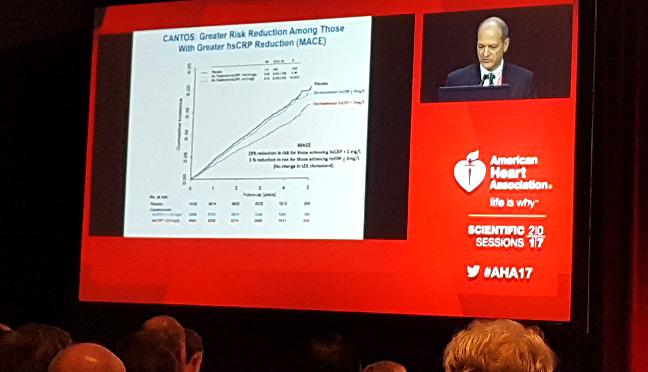
ANAHEIM, CA—(UPDATED) Individuals who robustly respond to treatment with canakinumab (Novartis) get the biggest cardiovascular bang for the buck, a new analysis from the CANTOS trial shows. Investigators, who presented the data here at the American Heart Association 2017 Scientific Sessions, showed that the magnitude of the reduction in high-sensitivity C-reactive protein (CRP) following a single dose of the drug can identify those with the largest reduction in major adverse cardiovascular events.
Among individuals with a previous MI and elevated levels of high-sensitivity CRP, those who achieved a CRP concentration of less than 2.0 mg/L on canakinumab, a fully human monoclonal antibody that targets inflammatory pathways, had a 25% reduction in the risk of nonfatal MI, nonfatal stroke, or cardiovascular death when compared with individuals treated with placebo.
Importantly, those who responded to treatment also had highly significant reductions in cardiovascular and all-cause mortality.
For those with an on-treatment high-sensitivity CRP concentration ≥ 2.0 mg/L, however, there was no reduction in the risk of major cardiovascular events, report investigators.
Paul Ridker, MD (Brigham and Women’s Hospital, Boston, MA), who led the secondary analysis of the CANTOS trial, told TCTMD that no matter which way they sliced the data, those who responded to canakinumab had significant reductions in adverse outcomes while those who didn’t respond gained very little from the still-investigational therapy.
“The biology is overwhelmingly telling us who responds to the drug,” said Ridker. “It’s pretty cool. We sliced it multiple ways, but most importantly, it didn’t matter. If you were in the group that responded—the group we’re calling robust responders—[you are] getting a 31% reduction in cardiovascular mortality, a 31% reduction in all-cause mortality, and 25% reduction in MACE. Highly significant, lots of zeros in the P value.”
For those who didn’t do as well on treatment, Ridker said there are still benefits, but the cardiovascular event reduction is very small. “There’s a biologic component of this that is fascinating,” he said. “The biology is actually driving the benefit.”
The new data were also published simultaneously in the Lancet.
Targeting the Inflammatory Pathway, Not LDL Cholesterol
Canakinumab targets interleukin-1β, a cytokine that is involved in the inflammatory response and the interleukin-6 signaling pathway. Interleukin-β is known to play “multiple roles in the development of atherothrombotic plaque,” according to the investigators. These roles include inducing procoagulant activity, promoting monocyte and leukocyte adhesion to vascular endothelial cells, and promoting the growth of vascular smooth-muscle cells.
As previously reported by TCTMD, the primary outcomes of CANTOS, which included 10,061 patients with a previous MI and high-sensitivity CRP levels ≥ 2 mg/dL (the median baseline CRP level exceeded 4.0 mg/L), were presented a few months back at the European Society of Cardiology Congress 2017 in Barcelona, Spain. Briefly, treatment with 150 mg of canakinumab every 3 months reduced the relative risk of nonfatal MI, nonfatal stroke, or cardiovascular death—the study’s primary composite endpoint—by 15% over median follow-up of 3.7 years when compared with placebo (P = 0.021).
In the new analysis, investigators sought to identify patients who responded to treatment with the fully human monoclonal antibody and whether on-treatment CRP concentrations predicted clinical outcomes. Of those given the 50-mg dose, 44% of patients achieved a CRP concentration of less than 2.0 mg/L. Comparatively, 55% and 65% of patients given the 150 mg and 300 mg doses, respectively, had similar reductions in CRP.
The number needed to treat (NNT) to prevent one MI, stroke, coronary revascularization, or death in the overall CANTOS trial is 24 at 5 years. However, the NNT is just 16 at 5 years among those with an on-treatment CRP concentration less than 2.0 mg/L, said Ridker. Among those with higher on-treatment CRP levels, the NNT is 57.
C. Michael Valentine, MD (Stroobants Cardiovascular Center/Centra Health, Lynchburg, VA), who was not involved in the study, said CANTOS “was a revelation to us,” as it identified a new anti-inflammatory agent to reduce cardiovascular events.
“But then you couple that with an injectable drug that will be expensive, hard to administer, and hard to get patients to comply with, that’s going to be the very difficult part of it,” Valentine, vice president of the American College of Cardiology, told TCTMD. “Very wisely, though, I think what they’ve done is asked how they can make it applicable to real-world scenarios and make it cost-effective.”
The ability to gauge which patients might best respond to therapy will prove important given the current cost of the agent, now approved for other, rarer, indications. In the United States, when given monthly for those approved indications, canakinumab is priced at approximately $200,000 per year. However, if approved for cardiovascular event reduction in secondary prevention, it would not likely be priced as high as that since it would lose orphan-drug status, said Ridker.
Valentine noted that most physicians don’t routinely measure CRP in post-MI patients or the secondary-prevention setting. “The question is whether we should be measuring it, to what effect are we lowering it, and what groups of patients stand to benefit from this,” he said.
Howard Weintraub, MD (NYU Langone Medical Center, New York, NY), told TCTMD that several analyses published to date have provided compelling evidence for lowering LDL cholesterol levels beyond traditional targets. While patients in CANTOS had a baseline LDL cholesterol level of 82 mg/dL, which is not exactly high, Weintraub said he’d aim to lower LDL cholesterol even further before thinking about inflammation.
“I’d definitely want more LDL reduction and would not think we’d gone as far as we could go,” said Weintraub. “And only then would I use canakinumab if was available and not prohibitively expensive.”
He added that the CANTOS study showed canakinumab was associated with a small but significantly increased risk of death caused by infection or sepsis when all three treatment arms were combined. Interestingly, fatal infection was lower in patients with CRP levels below 2.0 mg/L compared with those with higher CRP levels (0.27 vs 0.35 per 100 person-years), although rates were still higher compared with placebo
“I think what this study will encourage thoughtful physicians to do is get LDL cholesterol levels down and then look at CRP,” said Weintraub. “I’m grouping myself into the ‘thoughtful physician’ group, but I haven’t really been routinely measuring CRP because I didn’t find that it did anything to seriously alter my practice habits.”
As for what he considers an ideal LDL cholesterol level, Weintraub said he follows the advice of Michael Brown and Joseph Goldstein—the 1985 winners of the Nobel Prize for Physiology or Medicine—who suggested 25 mg/dL.
Is Lower CRP Better?
Speaking with the media, Ridker addressed whether individuals who achieve very low levels of CRP might achieve even greater reductions in cardiovascular events. He noted that when researchers stratified patients by achieved CRP levels, there was no significant reduction in the primary endpoint of major cardiovascular events among those in the highest tertile. Among those in the middle and bottom tertiles of achieved CRP concentrations, though, there was a statistically significant 17% and 29% reduction in risk, respectively, when compared with placebo-treated patients.
“Yes, I do think we have evidence, at least with canakinumab, that lower is better for inflammation reduction, analogous to how lower is better for LDL reduction,” said Ridker. “Interestingly, we’re getting a mortality reduction with that, which we haven’t seen with the PCSK9 inhibitor trials.”
Donald Lloyd-Jones, MD (Northwestern University Feinberg School of Medicine, Chicago, IL), who moderated the press conference, noted that reductions in inflammation can be achieved with smoking cessation, weight loss, and other lifestyle changes. If patients adopt these lifestyle modifications after starting treatment, this can positively affect clinical outcomes.
In terms of clinical practice, Ridker suggested the drug might be first prescribed as a single dose in eligible patients to identify responders. “I think it’s good medicine,” he said. “I measure blood pressure and if patients don’t respond, I change drugs. But cardiology has been very slow to adopt the idea that biology can drive the use. It’s also very important for the field in that the biologic response can tell you where the benefit is going to be.”
To TCTMD, Ridker said patients in CANTOS were extremely well treated with high-intensity statins, aspirin, and other medications. Despite the aggressive medical therapy, event rates in the placebo arm were high, he observed. “If you have this residual inflammatory problem, these drugs aren’t affecting that,” said Ridker. “Now we have evidence that the magnitude of the IL-6 reduction, the proxy being the CRP reduction, is really telling us who benefits [from canakinumab].”
Karol Watson, MD, PhD (University of California, Los Angeles, CA), who was not involved in the study, said the CANTOS trial confirms the long-standing belief that inflammation plays a role in atherosclerosis and cardiovascular events. “Finding an agent that can modulate inflammation that is tolerable and cost-effective is going to be essential to helping us stem the tide of atherosclerosis and cardiovascular mortality,” said Watson.
The Cardiovascular Inflammation Reduction Trial (CIRT), a 7,000-patient trial led by Ridker and sponsored by the National Heart, Lung, and Blood Institute, is also currently evaluating the role of inflammation in cardiovascular disease. In that study, which is enrolling patients with prior MI/documented coronary artery disease and type 2 diabetes or metabolic syndrome, low-dose methotrexate will be compared with placebo for the reduction of MI, stroke, and cardiovascular death.
Michael O’Riordan is the Managing Editor for TCTMD. He completed his undergraduate degrees at Queen’s University in Kingston, ON, and…
Read Full BioSources
Ridker PM, MacFayden JG, Everett BM, et al. Relationship of C-reactive protein reduction to cardiovascular event reduction following treatment with canakinumab: a secondary analysis from the CANTOS randomized controlled trial. Lancet. 2017;Epub ahead of print.
Disclosures
- Ridker received research grant support from Novartis Pharmaceuticals to conduct the CANTOS trial. He reports serving as a consultant to Novartis and is listed as a co-inventor on patents held by the Brigham and Women’s Hospital that relate to the use of inflammatory biomarkers in cardiovascular disease and diabetes that have been licensed to AstraZeneca and Siemens.


Comments