CT Perfusion Imaging for In-Stent Restenosis Shows Promise
ADVANTAGE, comparing the imaging vs CTA in stented patients with no prior MI, hints at “usefulness.” But questions remain.
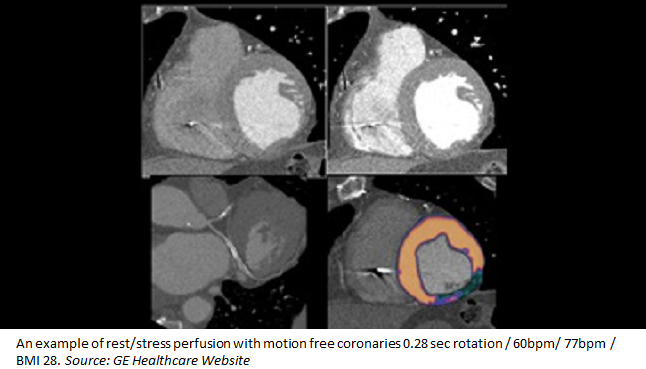
Stented patients are a known challenge for imagers: they often have a high atherosclerotic burden in nonstented segments and metallic stent structs can result in artifacts, which reduce the ability to make an accurate diagnosis. While stress myocardial CT perfusion can uniquely combine anatomical and functional evaluation, only recently has a novel whole-heart CT scanner been introduced that has shown promise even among patients with high heart rates.
“The ADVANTAGE study shows a potential usefulness of cardiac CT in the follow-up of patients with previous stent implantation,” write Daniele Andreini, MD, PhD (University of Milan, Italy), and colleagues. “The addition of a functional assessment with CT perfusion to the standard evaluation of de novo stenosis or in-stent restenosis by coronary CT angiography seems to overcome some drawbacks of cardiac CT in this clinical field, as a high rate of false positive and a weak correlation with noninvasive and invasive functional assessment.”
CT Perfusion Over CTA
Published online yesterday ahead of print in JACC: Cardiovascular Imaging, the ADVANTAGE study included 163 consecutive stable patients with coronary stents who were referred for ICA either because of chest pain and/or a positive stress test. Thirteen patients were excluded due to prior MI (n = 4), contraindication to adenosine administration (n = 4), impaired renal function (n = 3), and body mass index > 35 kg/m2 (n = 2). The remaining 150 underwent coronary CTA and stress myocardial CT perfusion using the aforementioned whole-heart scanner (Revolution CT; GE Healthcare), and all received ICA within the following 60 days.
In both territory-based and patient-based analyses, the researchers identified a higher proportion of evaluable patients for perfusion CT compared with coronary CTA (96.7% vs 91.1% and 96% vs 68%, respectively; P < 0.0001 for both).
Using ICA as a gold standard, the diagnostic accuracy of CT perfusion and coronary CTA were similar when only evaluable patients or territories were included, but CT perfusion outperformed coronary CTA when nonevaluable patients (86.7% vs 76.7%) and territories (92.1% vs 85.5%) were defined as positive (P < 0.03 for both). The combination of CT perfusion and coronary CTA resulted in the highest value of diagnostic accuracy (96.1% in the territory-based analysis).
In a separate FFR analysis of 36 patients, the diagnostic accuracy of coronary CTA fell to 35.5% when only evaluable vessels were included and 30.5% for all vessels, but that of CT perfusion stayed higher at 75% in both calculations (P ≤ 0.003 for both).
Andreini and colleagues state that these results “confirm the few data available in the literature” but included even more complex patients—the mean number of stents per patient here was 3.2. “Moreover, this study is the first assessment of the diagnostic accuracy of CT perfusion in stented patients not only versus ICA but also versus FFR, [which] is considered the functional gold standard,” the researchers say.
Additionally, they note, the findings were obtained with “very low” radiation exposure (4.15 ± 1.5 mSv for coronary CTA plus CT perfusion).
“Future studies, performed in asymptomatic populations and accompanied by outcome information, may provide further insights on the opportunity to include coronary CT angiography plus CT perfusion in the routine management of patients with previous coronary revascularization,” the authors conclude.
Moving Forward
In an accompanying editorial, Eike Nagel, MD, PhD (Goethe University Frankfurt, Germany), and Koen Nieman, MD, PhD (Stanford University School of Medicine, CA), explain that CT perfusion has struggled to find its place in clinical practice due to technical limitations requiring several heart beats to acquire a full picture. Additionally, it has been in “direct competition” with FFRCT, which does not require additional imaging beyond CT angiograms, and also perfusion cardiovascular magnetic resonance (CMR) imaging, which does not involve any radiation exposure.
J. Jane Cao, MD (St. Francis Hospital, Roslyn, NY), who was not involved in the study, told TCTMD that “CT perfusion is still widely considered as experimental, so there is no sort of reimbursement provided by Medicare or by other insurance companies.” Because of all this, “we're not ready for prime time [use of] CT perfusion,” she said.
Moreover, because the current analysis excluded patients with prior MI—something Cao said was necessary to determine the isolated diagnostic accuracy of CT perfusion over coronary CTA—these results cannot be applied to a real-world setting, where it would be precisely this population in which imagers are most challenged to identify new blockages or infarcts and who are at higher risk for them.
Nevertheless, Nagel and Nieman write that the study “sheds some light on the optimal use of imaging tests.” The three main messages, they say, are that more complex patients have a lower diagnostic yield with coronary CTA but cause fewer problems for perfusion imaging; the diagnostic accuracy of coronary CTA is lower in a patient population with a high prevalence of disease and even lower for those with intermediate stenosis when functional severity is the reference; and that noninvasive functional testing mimics invasive functional testing more so than anatomical imaging.
To translate the findings into clinical practice, the editorialists say it’s “obvious” that clinicians should opt for functional testing in those with known disease or a higher likelihood of CVD. But open questions remain, including whether coronary CTA provides any relevant additional information in patients with prior revascularization, how the results compare to CMR, how patients with a high pretest likelihood of disease but no prior stenting would do, and whether a diagnostic algorithm for patients with known CAD and chest pain could be created.
“A robust body of evidence has established the value of cardiac CT in patients without a prior history of coronary artery disease,” Nagel and Nieman conclude. “However, advanced CAD, myocardial scar, or prior revascularizations pose challenges to any diagnostic technique and more research will be needed to determine the effectiveness of cardiac CT to guide management in these complex patients.”
Cao said she would like to see future research on improving image postprocessing for CT perfusion. The example shown in the paper is “very fine quality” but hard to interpret without color coding. “Postprocessing development will really enhance our ability to differentiate areas with perfusion defect from the normal area,” she said, adding that this would “definitely help with the diagnostic accuracy.”
Lastly, she would like to see additional work on quantitative evaluation with perfusion imaging “to know the exact amount of blood flow going into the myocardium or the blood muscle so that we can be confident as to exactly what is normal and what is abnormal. . . . If we have quantitative levels of blood flow at the myocardium level, [this] will truly help to improve accuracy that is available for say cardiac MRI. It's the same direction where cardiac MR and PET imaging have been established for years and now CT is sort of the newcomer.”
Yael L. Maxwell is Senior Medical Journalist for TCTMD and Section Editor of TCTMD's Fellows Forum. She served as the inaugural…
Read Full BioSources
Andreini D, Mushtaq S, Pontone G, et al. CT perfusion versus coronary CT angiography in patients with suspected in-stent restenosis or CAD progression. J Am Coll Cardiol Img. 2019;Epub ahead of print.
Nagel E, Nieman K. Cardiac CT after coronary revascularization: enough to make a difference? J Am Coll Cardiol Img. 2019;Epub ahead of print.
Disclosures
- The study has received a grant from General Electric (GE Healthcare, Milwaukee, WI).
- Andreini reports serving on the speaker bureau for GE Healthcare and receiving institutional research grants from GE Healthcare and Bracco.
- Nagel reports receiving unrestricted institutional research support from Bayer AG.
- Nieman reports receiving unrestricted institutional research support from Siemens Healthineers, Bayer healthcare, GE Healthcare, and HeartFlow.
- Cao reports no relevant conflicts of interest.


Comments