Early Pilot Study Results Show Symptom Improvement With TMVR
It’s early days, say experts. For TMVR to take off, research will need to show better survival and improvements in other hard clinical outcomes.
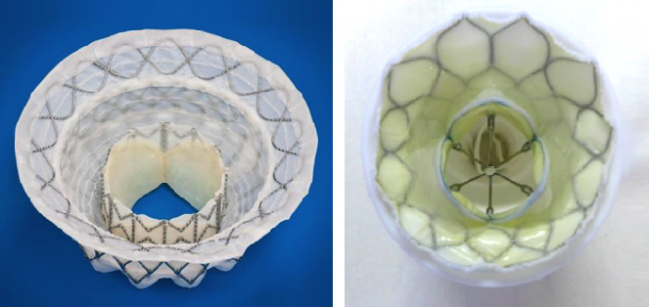
DENVER, CO—It’s early days, and the studies are small, but two new reports highlight the feasibility of transcatheter mitral valve replacement (TMVR) in patients with severe symptomatic mitral regurgitation (MR) who are considered poor candidates for surgery.
The two studies, named for the devices used—the Tendyne Global Feasibility Study and the Intrepid Global Pilot Study—were presented last week at TCT 2017 in Denver, CO. The Intrepid results were simultaneously published in the Journal of the American College of Cardiology. Both studies showed that transcatheter replacement of the mitral valve improved NYHA functional class, patient symptoms, and relief of MR, among other endpoints.
David W.M. Muller, MBBS, MD (St. Vincent’s Hospital, Sydney, Australia), who presented the 1-year outcomes on 30 patients treated with the Tendyne transcatheter mitral valve (Tendyne/Abbott), told TCTMD that for the field to advance patient selection will be critical.
“This is a small experience, as is clear, and the whole field still has relatively little experience,” said Muller. “We need a lot more effort to determine who is going to benefit from these procedures. From my input, I think it’s one thing to fix the mitral regurgitation but a lot of these people do have very severe left ventricular dysfunction and I think we will see a drop off—even though we fix the MR, we may not necessarily fix the patient long term. From the early experience, we are keeping them out of hospital and less symptomatic.”
Paul Sorajja, MD (Minneapolis Heart Institute, MN), who presented the 30-day results on 50 patients treated with the Intrepid TMVR system (Medtronic), said they are hopeful about the field, but there are challenges that need to be addressed.
“Certainly, the efficacy as compared with surgery, as well as safety, needs to be demonstrated, and the only way to do that is with a randomized trial,” said Sorajja.
INTREPID and TENDYNE
In the Intrepid pilot study, 50 patients with symptomatic, severe MR considered at high or extreme risk of death or complications from surgery were included. Overall, 36 patients had secondary MR, 8 patients had primary MR, and six had a combination of primary and secondary MR. Average LVEF was 43.4%. The Intrepid device, which is delivered transapically, was successfully implanted in 48 of 49 patients, with one procedure aborted.
In the first 30 days, seven patients died and five developed acute renal impairment. Five patients underwent reoperation for bleeding and four were rehospitalized for heart failure. There were no cases of device embolization or thromboses.
At 30 days, 74% of patients had no MR, with mild MR found in the remaining 26%. Additionally, 79% of patients at the last follow-up had NYHA class I or II heart failure compared with just 14% at baseline. In longer-term follow-up, an additional four patients died. All the deaths occurred before the 6-month mark, noted Sorajja.
With the Tendyne feasibility study, the transapically-delivered device was implanted in 27 patients with secondary MR and three with primary MR, of whom 17 individuals had an impaired LVEF. The vast majority of patients had severe (grade 4) regurgitation. The valve was successfully implanted in 28 patients.
At 1 year, five patients had died and three were rehospitalized for heart failure. In terms of valve performance, there was one case of malposition/hemolysis, one case of leaflet thrombosis, and one case of MR. Overall, 21 of 22 patients with 1-year follow-up had no MR symptoms and 19 of 20 patients improved to NYHA functional class I or II.
More Than Correcting MR
Commenting on the hurdles for the TMVR field, Sorajja said one important challenge will be getting heart-failure specialists and other physicians who care for these patients on board. He pointed out that in the vast majority of patients, MR is a ventricular disease, and that for TMVR to succeed, researchers will need to show more than a correction of MR.
“The predominant etiology of MR in these patients is secondary,” said Sorajja. “That’s where the biggest market is and that’s the biggest undertreated population. The heart-failure doctors are used to hard endpoints in their data, whether it’s a drug trial, a device trial, or an LVAD trial—they like survival and they like heart-failure hospitalizations to be reduced. Our trials in [TMVR] have to . . . demonstrate a benefit not just comparative to surgery, but above and beyond [existing] MR therapies.”
Just last week, Medtronic enrolled the first patient in the two-pronged APOLLO Transcatheter Mitral Valve Replacement pivotal study.
In total, 650 patients with severe, symptomatic MR who are candidates for mitral valve replacement surgery (and not candidates for mitral valve repair) will be randomized to receive the Intrepid device or undergo conventional surgery. The study is designed to show noninferiority between the two approaches at 1 year with respect to the primary endpoint of all-cause mortality, all-stroke, reoperation, and cardiovascular hospitalization.
To TCTMD, Sorajja said the main advantage with TMVR is avoiding cardiopulmonary bypass. “We’re not going to get too excited,” he said. “We’ll look at sternal wound infections, but that’s not going to be the [point] of Intrepid versus conventional surgery. It’s really going to be demonstrating noninferiority compared with surgery and achieving that without any potential adverse effects.”
In the single-arm cohort study that is part of APOLLO, 550 patients considered too high risk for conventional mitral valve surgery will be assigned to the TMVR procedure with the Intrepid system. The primary endpoint, which is the same as in the randomized trial, is a statistical noninferiority endpoint compared with a historical performance goal at 1 year.
Mayra Guerrero, MD (Evanston Hospital, IL), who was involved in the Tendyne feasibility study, told TCTMD she is cautiously optimistic about the TMVR field, noting that the different technologies, each with differing mechanisms of action, have shown the procedure can be done.
“Overall, early findings are encouraging, but refinements in techniques are needed, as well as better patient selection. There will be significant improvements in the near future,” said Guerrero. “This will mean treatment options for patients with limited options at this time.”
At this point, most transcatheter mitral valve repair options are transseptal, whereas most replacement options are transapical. If physicians could safely replace the mitral valve with the same low-risk profile of transseptal repair technologies, Guerrero said that would be preferable. Such technology, however, does not yet exist.
Some hints as to how this field will play out may come from the COAPT trial, which at long last has completed enrollment. Results are expected to be presented at TCT 2018. COAPT evaluated the MitraClip as a repair strategy against guideline-directed medical therapy in patients with secondary (functional) MR. According to Guerrero, the results will help shape future clinical trials in this field, since there is still no evidence that intervening in patients with MR secondary to left ventricular dysfunction will have an impact on hard outcomes.
“The problem is the ventricle, not the valve,” she said. “And if COAPT shows that intervening on the valve provides benefit to the patient, that would be a major breakthrough. There is a very large patient population with secondary MR. If the concept is true, then there’ll be further opportunities for transcatheter mitral valve repair and replacement. By the time COAPT results are available, several early feasibility [transcatheter mitral valve replacement] studies will be completed and ready for pivotal trials.”
Photo Credit: Photos extracted from Muller and Sorajja’s presentations, TCT 2017.
Michael O’Riordan is the Managing Editor for TCTMD. He completed his undergraduate degrees at Queen’s University in Kingston, ON, and…
Read Full BioSources
Bapat V, Rajagopal V, Meduri C, et al. Early experience with new transcatheter mitral valve replacement. J Am Coll Cardiol. 2017;Epub ahead of print.
Muller DW. Transcatheter mitral valve implantation for severe mitral regurgitation: the Tendyne global feasibility trial 1 year outcomes. Presented at: TCT 2017. November 1, 2017. Denver, CO.
Disclosures
- Sorajja reports receiving consulting fees/honoraria from Abbott Vascular, Boston Scientific, Edwards Lifesciences, Integer, and Medtronic; and grant support from Abbott Vascular, Boston Scientific, Edwards Lifesciences, and Medtronic.
- Muller reports receiving grant/research support from Tendyne and Medtronic; and consulting fees from Medtronic, Abbott, Boston Scientific, 4Tech, and Cephea.
- Guerrero reports receiving research/grant support from Edwards Lifesciences; consulting for Tendyne Holdings/Abbott; and serving on the speaker’s bureau for Abiomed.


Comments