Sentinel Cerebral Protection Device in TAVR ‘Promising’ but Trial Misses Primary Endpoint
Adjusted analyses saw a benefit for the filter device, which was also safe, prompting some to say use it, just in case.
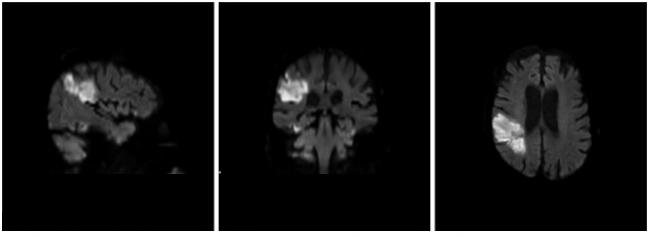
WASHINGTON, DC—Cerebral protection during transcatheter aortic valve replacement using the Sentinel filter device (Claret Medical) is safe and appears to trap embolic debris in almost every single procedure, but this does not translate into significant reductions in new brain lesion volume on MRI.
Those are the findings from the SENTINEL randomized trial, presented here at TCT 2016 by Susheel Kodali (NewYork-Presbyterian Hospital/Columbia University Medical Center New York, NY), and published online simultaneously with first author and co-principle investigator, Samir Kapadia, MD (Cleveland Clinic, OH), in the Journal of the American College of Cardiology.
“In high-risk patients undergoing TAVR, the Sentinel dual filter embolic protection device was associated with a favorable safety profile and . . . captured debris in almost all patients,” Kodali said in the Main Arena. However, he continued, “although there was a reduction in DW-MRI new lesion volume with embolic protection by 42%, the primary efficacy endpoint was not met.”
Preventing Neurologic Events in TAVR
Kodali, Kapadia, and colleagues from 18 US and European centers randomized 363 patients undergoing TAVR 2:1 to cerebral protection or no cerebral protection. Patients assigned to cerebral protection were further randomized to a “safety” group designed to look at MACCE at 30 days and an “imaging” group followed with MRI scans within the first week, with an endpoint of reduction in new lesion volume.
As Kapadia showed here, debris was found in 99% of cases and made up of thrombus with tissue elements, artery wall, calcification, valve tissue, and foreign materials. Its ubiquity is a reminder, say investigators, of the need to avoid whenever possible “overly aggressive device manipulation within the aortic valvar complex.”
MACCE rates in the cerebral protection group were noninferior to the performance goal and not statistically different from patients treated without the protection device (7.3% vs 9.9%; P = 0.41). Likewise, new lesion volume was numerically lower but not statistically different between the protection and no protection groups (102.8 mm3 vs 178.0 mm3, P = 0.33).
However, after adjusting for baseline lesion volume and valve type, there was a significant reduction in new lesion volume in protected territories (P = 0.02) with embolic protection.
More Embolic Debris According to Valve Type?
The valve data are of particular interest to experts in this space, since it’s possible that different valve profiles or delivery systems might be precipitating more neurocognitive adverse events. In SENTINEL there was a statistically significant difference in median new lesion volume between protection versus no protection seen for Sapien XT-treated patients and a numeric difference for the Evolut R-treated patients that didn’t meet statistical significance, but there was no difference for patients treated with Sapien 3.
The primary efficacy endpoint was not met.
Susheel Kodali
Kodali, speaking to the press, emphasized that while the analysis was underpowered, researchers did see a significant interaction between valve type and treatment arm. “The goal of this is not to say one valve type [causes more neurological infarcts than] another. There are confounders when you do any of these studies and there are interactions found,” he said, adding, “I agree these are small numbers. And we’re not trying to make conclusions on one valve type versus another, because there are purely baseline differences and there are physician choices as to why they used one valve type versus another, and you can’t account for that.”
During the Main Arena presentation, however, Kodali observed: “The reality is, these valves interact differently with the valve arch and the valve itself. This doesn’t answer the question of valve type, but clinically it makes sense that there would be different interactions with the valve annulus.”
Stroke Rates Post-TAVR
Stroke rates were also not significantly different, but of note were numerically higher in the control arm than what has previously been reported in observational trials (9.1 % vs 5.6%, P = 0.25). This detail prompted Alexandra Lansky, MD (Yale University, New Haven, CT), speaking with TCTMD, to note that it points to the fact that when followed rigorously, with neurologist review, stroke following TAVR may actually be more common than many physicians realize today.
Patients are going to say, ‘Well, then why wouldn’t you use a device like this? David R. Holmes Jr
Discussing stroke rates in the Main Arena, Kodali agreed that they do really depend on “who is doing the assessment.” A stroke rate of 9%, when a neurologist is doing the assessment, is likely within the bounds of what is seen in clinical practice, he said.
Panel discussant and study co-author Raj Makkar, MD (Cedars-Sinai Medical Center, Los Angeles, CA) pointed to the fact that SENTINEL investigators saw very few periprocedural strokes in the first 48 hours of TAVR, suggesting there is “a real impact” of cerebral protection that’s “worth investigating further.”
He added: “Despite nonsignificant P values, I think this is a very relevant finding.”
What Patients Want
Commenting on the findings during the morning press conference, David R. Holmes Jr, MD (Mayo Clinic, Rochester, MN), called cerebral protection “an incredibly important topic. There are several ways you can look at a study like this. You could look at a study like this statistically and say this didn’t meet its primary endpoint so everything is hypothesis generating, and that is true.”
Or, he continued, you can look at this from a patient standpoint.
“You can have a device that is safe, because the complication rate was very low, number one,” Holmes said. “Number two, we know it captures things that goes to your brain and it cannot be true that debris that goes to your brain is a good thing. So patients are going to say, ‘Well, then why wouldn’t you use a device like this?’”
Jeffrey Popma, MD (Harvard Clinical Research Institute, Boston, MA), was a little bit more cautious saying, “To use it, I would need to know what the success rate is for placement of the device and whether there are any procedure-related complications related to the device, how easy it is to use, how often you can use it, and whether there are any anatomic exclusion criteria on CT to be able to [use] it. So we need to delve into the depths of the details a bit. But I agree with Dr. Holmes that a reduction in lesion volume is a useful thing.”
In the late-breaking session, experts used words like “promising,” “intriguing,” and “hopeful.” But some, like Robert Bonow, MD (Northwestern University, Chicago, IL), said they want to see larger studies. “I’m very impressed,” he said. “This is very promising but thus far inconclusive. I’m leaning towards yes [about using it in patients today], but as an academic person as well as a person who sees patients, I’d like to see more data.”
Kodali, in the press conference, stressed that meta-analyses using the Sentinel device are forthcoming and the body of work on cerebral protection supports its safety and benefits—a point also made in an accompanying editorial by Azeem Latib, MD, and Matteo Pagnesi, MD (San Raffaele Scientific Institute, Milan, Italy), in JACC.
Embolic protection is widely used in carotid stenting, Kodali continued, despite the fact that no randomized trials have proven long-term clinical benefits. Such a trial, he noted, could now likely not be ethically performed.
In fact, Claret Medical has already filed for FDA clearance of the Sentinel device.
Photo Credit: Michael Dwyer, Buffalo Neuroimaging Analysis Center
Shelley Wood was the Editor-in-Chief of TCTMD and the Editorial Director at the Cardiovascular Research Foundation (CRF) from October 2015…
Read Full BioSources
Kapadia SR, Kodali S, Makkar R, et al. Cerebral embolic protection during transcatheter aortic valve replacement. J Am Coll Cardiol. 2016;Epub ahead of print.
Latib A, Pagnesi M. Cerebral embolic protection during transcatheter aortic valve replacement: a disconnect between logic and data? J Am Coll Cardiol. 2016;Epub ahead of print.
Disclosures
- Kodali reports being a consultant for Edwards Lifesciences. Disclosures for co-authors are listed in the paper.
- The editorialists disclose institutional grants for clinical studies of cerebral embolic protection devices developed by Claret Medical, Keystone Heart, and Innovative Cardiovascular Solutions.


Comments