Studies Zero in on Delayed Conduction Problems After TAVI
Experts agree that extended ambulatory ECG monitoring should be more widely used after TAVI, especially in high-risk patients.
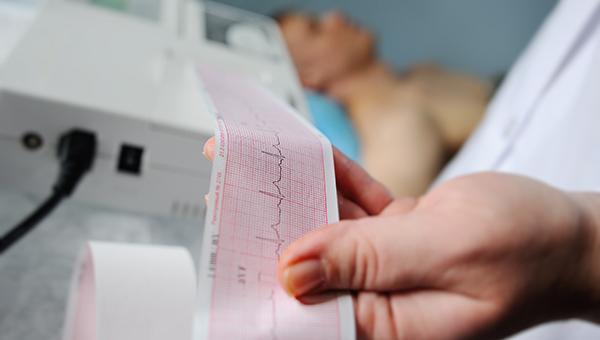
Delayed cases of total atrioventricular block (AVB) or complete heart block can occur more than 2 days after TAVI, two new studies affirm, highlighting the utility of extended ECG monitoring after discharge, particularly in patients with certain high-risk features.
In one study, 4.6% of patients had delayed high-degree AVB or complete heart block detected with 14 days of ambulatory ECG monitoring, and in the other, 11.9% of patients had delayed total AVB picked up by 1 year of monitoring with an implantable loop recorder (ILR).
The development of new conduction disturbances leading to permanent pacemaker implantation is a relatively frequent complication of TAVI, although the best way to manage the problem remains unclear. Data are especially limited when it comes to conduction disturbances that occur more than 2 days after the procedure.
These two new studies, published ahead of print in the December 27, 2021, issue of JACC: Cardiovascular Interventions, provide some insights into the frequency of delayed AVB or complete heart block after TAVI, the features that place patients most at risk, and the potential role of extended ECG monitoring.
Summing up the findings for TCTMD, Scott Lilly, MD, PhD (The Ohio State University Wexner Medical Center, Columbus), who was the lead author of the 2020 American College of Cardiology expert consensus decision pathway on the management of conduction disturbances in patients undergoing TAVI, said, “Ambulatory monitoring after TAVR should be broadly utilized, particularly in high-risk patients.”
Josep Rodés-Cabau, MD, PhD (Quebec Heart and Lung Institute, Laval University), senior author of the study that used ambulatory ECG monitoring, agreed. A larger study, ideally randomized, is still needed to confirm the utility of the approach, but in the meantime, “it seems to be worth it to have some kind of monitoring for patients with high-risk features,” he told TCTMD. He pointed to ECG changes that occur during TAVI as a way to identify patients who should be evaluated for a short time after discharge, as shown by both studies.
Baseline RBBB, New Conduction Disturbances Increase Risk
In the first study, Guillem Muntané-Carol, MD (Quebec Heart and Lung Institute, Laval University, Canada), Rodés-Cabau, and colleagues explored the use of ambulatory ECG monitoring following TAVI performed with a minimalist approach to look for delayed conduction disturbances. The analysis included 459 patients (mean age 79; 45.3% women) who did not have a permanent pacemaker and who underwent continuous rhythm monitoring for 14 days after discharge, an approach suggested in an expert consensus document published in August 2019. Monitoring was performed using two ECG patches—CardioSTAT (Icentia) and Zio AT (iRhythm Technologies).
The researchers divided the patients into three groups:
- Group 1: no right bundle branch block (RBBB) and no ECG changes (68.6%)
- Group 2: baseline RBBB and no ECG changes (8.3%)
- Group 3: new-onset ECG conduction disturbances (23.1%)
Overall, delayed high-degree AVB or complete heart block were seen in roughly one out of every 20 patients on ambulatory ECG monitoring; of the affected patients, 81.0% had a permanent pacemaker implanted.
These disturbances were relatively rare in group 1 (2.2%), with higher risks in patients with baseline RBBB (13.2%) and in those with new-onset conduction disturbances after the procedure (8.5%). There were no sudden cardiac deaths—or any deaths for that matter—through 30 days.
“These results would support tailored management using ambulatory ECG monitoring and the possibility of longer hospitalization periods in patients at higher risk for delayed high-degree AVB or complete heart block,” the investigators write.
Risk Over the Longer Term
Christian Reiter, MD (Kepler University Hospital, Johannes Kepler University Linz, Austria), and colleagues performed a similar study among 59 patients (mean age 80.3; 39.0%) treated with TAVI at a single center. In this case, all patients also underwent invasive electrophysiologic (EP) testing and were fitted with an ILR (Reveal LINQ; Medtronic) for 1 year after remaining free from persistent or recurrent total AVB for the first 48 hours after the procedure.
This type of extended monitoring identified 11.9% of patients with delayed total AVB, with onset within the first 8 days after TAVI in five of seven affected patients. The remaining two patients developed the disturbance more than 3 months after the procedure. Of note, five of seven cases were detected only with the ILR and not on a surface ECG.
Factors associated with a greater likelihood of finding the disturbance were prolongation of the PQ interval between baseline and day 2 after TAVI (OR 1.04; 95% CI 1.01-1.09 per 1-ms increment) and prolongation of the HV interval during the procedure (OR 1.07; 95% CI 1.02-1.14 per 1-ms increment).
“Our findings suggest that personnel taking care of TAVR patients should screen and be alert for bradyarrhythmia and its associated signs and symptoms,” the authors say. “The regular use of ambulatory monitoring devices such as wearable ECG patch monitors, preferably linked to telemetric systems, should be considered for up to 14 days after TAVR. These devices could play an important role in the detection of delayed total AVB, especially in patients with risk factors for its occurrence.”
Extended ECG Monitoring for All TAVI Patients?
Asked whether all TAVI patients should receive extended ECG monitoring, Reiter said no, adding that he “would suggest a tailored approach in patients with high-risk ECG findings like RBBB or a significant increase of the PQ interval in the first days after TAVR. As most delayed total AVBs occurred in the first weeks after the procedure, and since ILR implantation is not cost-effective, using ambulatory ECG monitors (eg, wearable ECG patch monitors) for 2 to 3 weeks seems to be an adequate option.”
Regarding the factors found on invasive EP testing that were associated with greater risk of delayed total AVB, Reiter said the findings should be confirmed in larger trials. “As evaluation of the intraprocedural increase of the HV interval is a feasible, safe, and uncomplicated procedure and the increase of the HV interval significantly predicted new-onset of delayed total AVB in our patient cohort, implementation of this procedure in patients undergoing TAVR might have potential benefit in identifying patients at high risk for the occurrence of delayed total AVB or who should have extended ECG monitoring,” he said.
Currently, Lilly said, use of extended ECG monitoring after TAVI varies across centers. But based on these and prior studies, “it’s absolutely reasonable—and I would argue, appropriate—to do ambulatory monitoring on any patient that has a new conduction disturbance after TAVR, and our feeling is that should continue for at least 14 days and up to 30 days,” he said, noting that his center has been adhering to this approach for about two-and-a-half years.
One could argue that the same strategy should be applied to patients who have RBBB at the time of TAVI, regardless of whether it changes after the valve is implanted, an approach that would have as many as half of all patients being monitored after discharge, Lilly said. Devices for monitoring, however, “are not perfect,” he said, pointing to issues involving patient adherence, communication of ECG findings to the care team, reimbursement, and costs to patients.
If a monitor can provide information on conduction disturbances in real time to the care team, “then it allows you to dispatch EMS or to call the patient or to get to them before there are untoward consequences,” Lilly said, adding that findings provided days later are less actionable. “So not only does there need to be a standardization of practices [around extended ECG monitoring], but there needs to be standardization of features appropriate for monitoring these patients.”
Rodés-Cabau noted that it remains to be proven that performing ambulatory ECG monitoring in high-risk patients after TAVI protects against sudden death, although in general it’s believed that complete heart block is tied to a higher risk of more-serious events in the future. “Meaning that diagnosing these events, even if they are silent, probably protects you against more-severe events like syncope or sudden death in the end,” he said.
In an accompanying editorial, Mohamad Alkhouli, MD (Mayo Clinic, Rochester, MN), takes a broad view of the issue. “At the present time, selective short-term monitoring will likely continue to be the dominant strategy to address delayed high-degree AVB or complete heart block after TAVR,” he writes.
“However,” he continues, “futuristic innovations in smart devices, mini-sensors, and removable or dissolvable pacemakers are expected to move the field toward a ‘long-term monitoring for all, prophylactic pacemaker implantation for some’ strategy to address the growing concerns about delayed and very delayed conduction disturbances after the procedure.”
Todd Neale is the Associate News Editor for TCTMD and a Senior Medical Journalist. He got his start in journalism at …
Read Full BioSources
Muntané-Carol G, Okoh AK, Chen G, et al. Ambulatory electrocardiographic monitoring following minimalist transcatheter aortic valve replacement. J Am Coll Cardiol Intv. 2021;14:2711-2722.
Reiter C, Lambert T, Kellermair J, et al. Delayed total atrioventricular block after transcatheter aortic valve replacement assessed by implantable loop recorders. J Am Coll Cardiol Intv. 2021;14:2723-2732.
Alkhouli M. Delayed atrioventricular block after transcatheter aortic valve replacement: the new Achilles’ heel? J Am Coll Cardiol Intv. 2021;14:2733-2737.
Disclosures
- Muntané-Carol reports support by a grant from Fundación Alfonso Martin Escudero (Madrid, Spain).
- Rodés-Cabau holds the Research Chair “Fondation Famille Jacques Larivière” for the Development of Structural Heart Disease Interventions; and reports receiving institutional research grants from and serving as a consultant for Edwards Lifesciences, Medtronic, and Boston Scientific.
- Reiter reports no relevant conflicts of interest.



Comments