Baroreflex Activation for Advanced HF Yields Positive Results: Postmarketing Data
Even as BAT expands beyond trial criteria, patients see gains in LVEF, symptoms, and more. A survival impact remains unclear.

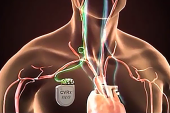
BOSTON, MA—Baroreflex activation therapy (BAT) delivered by the Barostim implant (CVRx) is associated with a range of benefits in patients who have advanced heart failure with reduced ejection fraction (HFrEF), according to postmarketing data presented here at THT 2026.
In the REBALANCE registry, patients saw improvements in LVEF and NYHA functional status 6 months after implantation, which reflects results of about 3 months of fully titrated therapy, Dmitry Yaranov, MD (Baptist Medical Group, Memphis, TN), reported. Longer-term data on a smaller number of patients indicated that the gains were sustained to 2 years.
A separate analysis of a large multi-institutional database, presented at THT by Arvind Bhimaraj, MD (Houston Methodist, TX), suggested that BAT may reduce all-cause hospitalizations and HF-related hospitalizations and emergency department (ED) visits.
Commenting on the REBALANCE registry results, Shaline Rao, MD (NYU Langone Health, New York, NY), told TCTMD it’s useful information “because now you’re mapping out this device to a real-world population that comes with many elements you can’t control.”
In fact, 33.6% of patients treated in REBALANCE received BAT off-label, defined as not meeting one of the following approved criteria that came from the BeAT-HF trial: LVEF ≤ 35%, NYHA class III symptoms (or class II symptoms with recent class III symptoms), and an NT-proBNP level < 1,600 pg/mL. The patients in the registry tended to be older, were more likely to be taking angiotensin receptor-neprilysin inhibitors (ARNIs) and sodium-glucose cotransporter 2 (SGLT2) inhibitors, and had more heterogeneous disease severity compared with the trial participants.
With the registry results placed in that context, “you are validating that there is value outside of the very highly controlled trial environment,” Rao said. After the BeAT-HF results came out, she said, many clinicians noted that it would be a challenge to find patients who adhered tightly to the profile of a trial participant.
One gap in knowledge that remains, however, is whether BAT induces a meaningful change in long-term survival, Rao added. “That’s an area that we’re waiting to understand more.”
The REBALANCE Registry
Abnormal baroreflex signaling is an initial step in the activation of neurohormonal pathways that are responsible for HF progression, Yaranov explained. Neurohormonal blockade is the basis of guideline-directed medical therapy (GDMT) to improve outcomes in patients with HF, and “baroreflex activation therapy can complement drug therapy by acting upstream to restore baroreflex signaling in those patients,” he said. That rebalances the autonomic nervous system by decreasing sympathetic activation and increasing parasympathetic activation, which in turn reduces HF symptoms.
The US Food and Drug Administration approved Barostim in 2019 on the basis of results from BeAT-HF, which showed that BAT improved quality of life and exercise capacity and kept 94% of patients free of major adverse neurological and cardiovascular events through 6 months.
The REBALANCE registry enrolled 435 patients (mean age 68 years; 69.8% men) at 45 US sites after approval of Barostim. Patients in the registry had increased use of GDMT compared with BeAT-HF participants, with 61.5% receiving an ARNI and 49.4% receiving an SGLT2 inhibitor; they were on a mean of 4.1 HF medications.
At baseline, mean LVEF was 26.8%. Most patients (74.1%) had NYHA class III symptoms and 20.7% had class II symptoms. Mean NT-proBNP was 929 pg/mL.
Six months after implantation of Barostim, there were no significant changes in NT-proBNP, renal function, systolic or diastolic blood pressure, or heart rate.
There were, however, significant increases in mean LVEF (to 29.9%) and NYHA class, with 34% of patients improving by at least one class (P < 0.001 for both).
Analyses in a smaller number of patients who completed follow-up beyond 6 months showed that mean LVEF increased to 30.8% at 1 year and 33.7% at 2 years, with similar findings in both the overall cohort and the subset of patients with on-label indications. Nearly half of patients (49.8%) saw a clinically meaningful rise in LVEF of at least 5%.
Improvements in NYHA class persisted out to 2 years as well.
“This multicenter, postmarket registry demonstrates sustained improvement in both LVEF and functional status in HFrEF patients after receiving Barostim [in] both the full REBALANCE cohort and on-label cohort,” Yaranov said.
In the study Bhimaraj reported, which relied on an Epic-based dataset called COSMOS, there were 1,533 patients (median age 69 years; 74% men) who received Barostim implants. Median NT-proBNP at baseline was 1,166 pg/mL, with about one-quarter of patients having a level above 1,600 pg/mL.
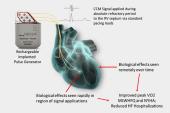
Despite the cohort having a high burden of comorbidities, there was a decline in NT-proBNP levels in the year following device implantation. When comparing the year after implantation with the year before, there was a 14.4% reduction in all-cause hospitalizations, a 29.1% drop in HF-related hospitalizations, and a 26.8% fall in HF-related ED visits. An 18.3% increase in all-cause ED visits was attributed to overall poor health of the cohort.
‘A Safe Tool’
Rao said patients do feel better with BAT, both in her clinical experience and in studies. The improvements in LVEF, walking distance, and other measures should translate into gains in survival, although that hasn’t been borne out in research studies yet, she added.
“I would be cautious and I wouldn’t counsel patients that it’s a guarantee that they will have any kind of change in their long-term survival,” she said.
But survival is not the only important outcome, she indicated. “For someone who is in an advanced heart disease scenario, who has a lot of day-to-day symptoms and functional capacity limitations, and who has hit a limit on how many medications they can take, there is still value in this because we are changing their other parameters that bring them value and improve their quality of life.”
Many patients with advanced HFrEF already have other implanted devices, and that’s something to consider when thinking about using Barostim, Rao noted.
Nonetheless, “we know that it’s safe and that it can improve walking times and quality of life in patients who have progressive [HFrEF] despite medical therapy and that even in an off-label population, somebody who doesn’t perfectly fit, there was value added,” Rao said. In patients who have progressive HF and are running out of drug and device options to improve their situation, she added, “this is a safe tool to consider.”
Todd Neale is the Associate News Editor for TCTMD and a Senior Medical Journalist. He got his start in journalism at …
Read Full BioSources
Yaranov D. Initial 6 month outcomes with baroreflex activation therapy from the REBALANCE registry. Presented at: THT 2026. March 2, 2026. Boston, MA.
Yaranov D. LVEF and functional improvement observed in a post-market registry of Barostim. Presented at: THT 2026. March 3, 2026. Boston, MA.
Bhimaraj A. Real-world outcomes of Barostim baroreflex activation therapy in heart failure: analysis of a large multi-institutional database. Presented at: THT 2026. March 3, 2026. Boston, MA.
Disclosures
- The REBALANCE registry is supported by CVRx.
- Yaranov reports consulting fees/honoraria from Abbott, AstraZeneca, Boehringer Ingelheim, Alnylam, BridgeBio, and CVRx.
- The study presented by Bhimaraj was supported by a principal investigator-initiated grant from CVRx.
- Bhimaraj reports receiving grant/research support from CVRx and Cardiol Therapeutics; receiving consulting fees/honoraria from Abbott and Abiomed; having stocks/options from Rhythm Science; and serving on advisory boards for Alnylam, Pfizer, and BridgeBio.



Comments