Intravascular Lithotripsy Shows Good Safety at 30 Days: DISRUPT CAD II
The approach permits successful lesion preparation in PCI patients with severe calcification, but longer follow-up is needed.
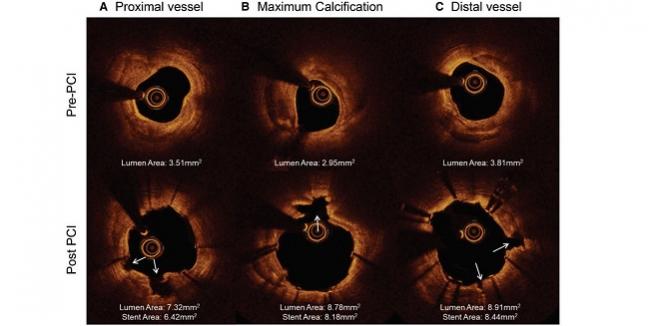
SAN FRANCISCO, CA—Intravascular lithotripsy used to fracture calcium in highly calcified arteries prior to PCI is safe, a small study confirms. The data also offer insights into the mechanism of action for this novel approach.
“We have demonstrated excellent safety in more centers and more patients with this novel technology,” said Carlo Di Mario, MD, PhD (Careggi University Hospital, Florence, Italy), who presented the primary findings of DISRUPT CAD II at TCT 2019.
Simultaneously published in Circulation: Cardiovascular Interventions, results follow the 60-patient DISRUPT CAD I study, which established the feasibility of lithotripsy in this setting. The Shockwave IVL system (Shockwave Medical; Fremont, CA) is made up of a semicompliant balloon catheter, connector cable, and generator; together, these deliver pulsatile mechanical energy that causes microfractures in the calcium. The therapy has shown promise in other initial studies but has yet to be compared head-to-head with other plaque modification methods like rotational and orbital atherectomy.
Low MACE With No Procedural Complications
For DISRUPT CAD II, Di Mario along with lead author Ziad Ali, MD, DPhil (NewYork-Presbyterian/Columbia University Irving Medical Center, New York, NY), and colleagues enrolled 120 patients with coronary artery calcium slated to undergo PCI at 15 hospitals in nine countries between May 2018 and March 2019. About one-third of patients were diabetic, and 8% were in renal failure. Overall, 94.2% of lesions were considered to have severe calcification on angiography, with an average 26 mm of calcified length and about 70% graded as concentric.
All patients received successful delivery and use of the lithotripsy catheter, and the postprocedure angiographic acute luminal gain was 0.83 mm. Residual stenosis was 32.7% and further decreased to 7.8% after DES implantation.
In-hospital MACE, the primary endpoint defined as cardiac death, MI, or TVR, occurred in 5.8% of patients, consisting of seven non–Q-wave MIs. There were no instances of procedural abrupt closure, slow or no reflow, or perforations. MACE at 30 days was 7.6%, including one cardiac death, one Q-wave MI, one TVR, and two definite/probable stent thromboses.
Longer-term Follow-up Needed
In a panel discussion following the presentation, Robert Gil, PhD (Central Hospital of the Ministry of the Interior, Warsaw, Poland), commented that “we all know that high-speed rotablation is not enough for [certain] lesions, especially in deep locations,” suggesting a potential need for this kind of lesion preparation. “The only bad point of this technology is the price, which is very high,” he said.
 Session co-moderator Dimitrios Alexopoulos, MD (Attikon University Hospital, Athens, Greece), expressed surprise at the safety shown in the study, especially given the limited experience available with this technology. “No complications, is this really true?” he asked. “It's difficult to accept that disruption of calcium will occur in every case in a predicted way and there will be no case of perforation or something like that. There are isolated cases published that there were some problems. Do you suggest it’s a matter of taking special precaution or is it a technique that is demanding?”
Session co-moderator Dimitrios Alexopoulos, MD (Attikon University Hospital, Athens, Greece), expressed surprise at the safety shown in the study, especially given the limited experience available with this technology. “No complications, is this really true?” he asked. “It's difficult to accept that disruption of calcium will occur in every case in a predicted way and there will be no case of perforation or something like that. There are isolated cases published that there were some problems. Do you suggest it’s a matter of taking special precaution or is it a technique that is demanding?”
Di Mario, who was also a PI on DISRUPT CAD I, assured that all patients were monitored and “we did not have [an] event that was not reported.” Experience has grown with the procedure, he continued, noting that “the main challenge is to deliver a balloon that, especially in the initial series, was fairly thick and relatively inflexible.”
The good safety seen in the study is related to several factors, Di Mario suggested. First, there is zero embolization. Secondly, “you can protect all the side branches you want because the wire can remain in place.” Lastly, he observed, “the mechanism is not like when you have calcified region to go to 40-50 atm and create an enormous dissection that clogs all branches and forces you to use longer stents than the calcified lesion itself, but you can stick simply to the diseased segment.”

But longer-term follow-up of this cohort is crucially needed, according to panelist Azfar Zaman, MD (Freeman Hospital, Newcastle upon Tyne, England). “The procedural outcomes are very good, but do we not have a duty to follow all these patients . . . to see what the long-term outcomes are?” he asked, noting that “[OCT images show] fracture of the calcium, but some of them show fracture beyond and possibly into the tunica media.”
“I agree it will be nice to have a series of patients with an angiographic follow-up to make sure that they have no aneurysm,” Di Mario said, adding that he’s never seen any in his 3.5 years of experience.
Photo Credit: Carlo Di Mario
Yael L. Maxwell is Senior Medical Journalist for TCTMD and Section Editor of TCTMD's Fellows Forum. She served as the inaugural…
Read Full BioSources
Ali ZA, Nef H, Escaned J, et al. Safety and effectiveness of coronary intravascular lithotripsy for treatment of severely calcified coronary stenoses: the Disrupt CAD II Study. Circ Cardiovasc Interv. 2019;Epub ahead of print.
Disclosures
- This study was funded by Shockwave Medical.
- Ali reports receiving personal fees and holding equity in Shockwave Medical; grants from Cardiovascular Systems and Abbott Vascular; and personal fees from Boston Scientific, AstraZeneca, ACIST Medical, Opsens Medical, and Cardinal Health.
- Di Mario reports receiving institutional grants from the Disrupt CAD studies from Shockwave Medical.


Comments