KARDINAL: Monthly Tonlamarsen May Not Enhance BP Lowering in Resistant Hypertension
Repeat injections of the angiotensinogen-targeted drug didn’t reduce BP more than a single dose.
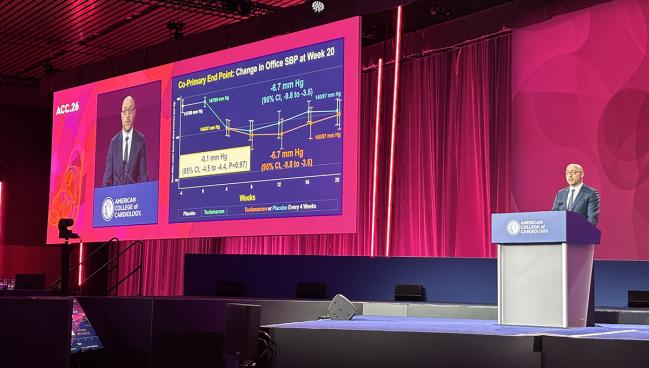
NEW ORLEANS, LA—For patients with uncontrolled hypertension already on two or more medications, monthly injections of tonlamarsen (Kardigan) more effectively lowered plasma angiotensinogen (AGT) levels than a single dose of the novel drug, according to results from the phase II KARDINAL trial. But repeat dosing did not lead to greater reductions in blood pressure over 20 weeks.
Luke J. Laffin, MD (Cleveland Clinic Foundation, OH), presented the results today at the American College of Cardiology (ACC) 2026 Scientific Session.
Tonlamarsen “suppresses hepatic angiotensinogen production,” Laffin told the media ahead of the late-breaking session. “We know that AGT suppression reduces blood pressure among patients taking zero to one medication. However, more data are needed in patients taking multiple medications, including ACE inhibitors and angiotensin receptor blockers.”
AGT is a precursor for all peptides generated in the renin-angiotensin-aldosterone system (RAAS).
Daniel W. Jones, MD (University of Mississippi, Jacksonville), a past president of the American Heart Association and writing committee chair for the 2025 US hypertension guidelines, cautioned that KARDINAL is a phase II trial that represents the early stages of investigation on tonlamarsen.
“It’s too early to really understand how and if this drug fits into clinical management of hypertension,” he told TCTMD in an email. “Similar to a few other investigational drugs, it might offer the potential for periodic drug injection as opposed to daily oral use to improve adherence. Patients are more likely to adhere to treatment plans when they are easier to follow; therefore, periodic injections may be preferred versus remembering to take a daily pill.”
Several additional drugs, as reported by TCTMD, are being explored as solutions to the vexing problem of treatment-resistant hypertension.
Zilebesiran (Alnylam Pharmaceuticals), a small interfering RNA agent that, like tonlamarsen, targets AGT production, has previously shown promise in the KARDIA-2 trial. Taking a different approach is lorundrostat (Mineralys Therapeutics), a novel aldosterone synthase inhibitor studied in the Advance-HTN trial. Another aldosterone synthase inhibitor, baxdrostat (CinCor Pharma) failed to show a benefit over placebo in the phase II HALO trial but did do so previously in BrigHTN. Additionally, there’s aprocitentan (Tryvio; Idorsia Pharmaceuticals), an endothelin receptor antagonist that was approved by the US Food and Drug Administration in 2024 for uncontrolled hypertension.
The KARDINAL Trial
KARDINAL enrolled adults with an office systolic BP greater than 135 mm Hg despite already receiving two to five antihypertensive drugs. More than 80% were on an ACE inhibitor or ARB. All had an eGFR greater than 30 mL/min/1.73 m2.
The mean age among randomized patients was 61 years, 59% were women, 33% had diabetes, 11% had an eGFR > 60 mL/min/1.73 m2, and 49% were Black.
The study involved three phases that started with a 4-week placebo lead-in period (n = 279), followed by a 4-week active run-in period during which patients (n = 206) received a single subcutaneous 90-mg injection of tonlamarsen. Following these 8 weeks, participants (n = 198) were randomized to receive either four additional tonlamarsen doses or matching placebo for 16 weeks.
KARDINAL had two primary endpoints: change from baseline to week 20 in plasma AGT and in clinic-measured systolic BP levels.
The mean in-office BP was 147/90 mm Hg before the placebo lead-in and 147/89 mm Hg after. After the active run-in period, mean BP was 140/87 mm Hg. Twenty weeks after the first tonlamarsen dose, the least-squares (LS) mean change in office systolic BP was nearly identical between the single-dose group (-6.7 mm Hg; 95% CI -9.8 to -3.5) and the monthly-dose group (-6.7 mm Hg; 95% CI -9.8 to -3.6; P = 0.97).
There was an advantage for monthly dosing when it came to plasma AGT. By week 20, patients randomized to a single tonlamarsen dose had an LS mean percent change of -23.0% (95% CI -27.8 to -18.2%), as compared to -67.2% (95% CI -71.9 to -62.4%) with monthly dosing (P < 0.0001).
Serious adverse events occurred in 2.1% of the single-dose group and 5.0% of the monthly-dose group, with the latter also experiencing more injection-site reactions. One patient receiving monthly tonlamarsen died, though the site investigator deemed the death unrelated to the study drug.
A New Target?
“Nucleic acid-based therapeutics decreasing hepatic AGT synthesis represent a new approach to RAAS modulation with drugs that are durable and not dependent on daily adherence,” the investigators note in their paper. “Hepatic AGT suppression targets the dominant source of circulating AGT and may provide more sustained reduction across dosing intervals in the downstream products of AGT, such as angiotensin II.”
KARDINAL, unfortunately, does not provide a clear answer on whether targeting AGT can ultimately lead to lower blood pressure for patients already on several antihypertensive medications, they say. The continued suppression of AGT 20 weeks after the single tonlamarsen dose was unanticipated given that the drug has an elimination half life between 2 and 4 weeks.
Morris Brown, MD (Queen Mary University of London, England), the senior investigator for the BrigHTN trial of baxdrostat, told TCTMD that he’s not convinced that it makes sense to target AGT in patients already on an ACE inhibitor and/or ARB, given that those drugs already target the RAAS. What AGT suppression might offer beyond these conventional therapies isn’t so much BP-lowering, he said, but rather the “possible convenience of only having to deliver the drug,” in this case monthly.
Additionally, “since angiotensinogen levels are not the rate-limiting step in determining the level of angiotensin II (the active molecule) in most patients, it is likely that near 100% reductions are required to achieve an effect on BP. We have known this for other components of the renin-angiotensin system—effective doses of an ACE inhibitor reduce enzyme activity by near 100%,” Brown explained.
Laffin suggested several reasons for the lack of difference in BP in KARDINAL.
“The first is that a single dose of tonlamarsen may result in sustained office blood pressure reduction, even as angiotensinogen suppression remains. The second is that residual AGT suppression may have attenuated the between-treatment-group differences in office blood pressure, [thus] requiring larger studies in larger sample size. And finally, he added, “it can’t be excluded, although I think this is less likely, that improved adherence to background medications or additional regression to the mean in blood pressure occurred following the placebo lead-in.”
A limitation of the trial, said Laffin, is its lack of a placebo-only comparator arm.
John D. Bucheit, PharmD (Virginia Commonwealth University, Richmond), a member of the ACC Prevention Council who took part in the press conference, agreed, but said the trial nevertheless looks at a key clinical need.
“Hypertension is a major problem that we all see, and our control rates in the US are less than what we’d like. So having novel treatment options is really important,” said Bucheit. The take-home here is that “this medication definitely reduces angiotensinogen, and so this is a potential target in therapy. What I think is important going forward is we are going to need those placebo-controlled arms.”
To TCTMD, Jones pointed out that KARDINAL’s design featuring a 4-week active run-in period is not common and “might make it more difficult to evaluate BP-lowering effectiveness.” The area most ripe for research, he added, is understanding what factors might be causing the resistance to treatment.
Caitlin E. Cox is Executive Editor of TCTMD and Associate Director, Editorial Content at the Cardiovascular Research Foundation. She produces the…
Read Full BioSources
Laffin LJ, Wang Q, Sarraju A, et al. Efficacy of tonlamarsen in patients with uncontrolled hypertension: the KARDINAL phase 2 randomized clinical trial. JACC. 2026;Epub ahead of print.
Disclosures
- KARDINAL was funded by Kardigan.
- Laffin reports serving as a consultant or on steering committees for Recor, Medtronic, Eli Lilly, Mineralys, Crispr Therapeutics, Idorsia, Novartis, Kardigan, Novo Nordisk, AstraZeneca, Boehringer Ingelheim, Arrowhead Pharmaceuticals, Ripple Medical, and Stability Health. He has received research funding from AstraZeneca for studies of rosuvastatin. He receives royalties from Belvoir Media Group, Elsevier, and Springer Nature. No personal renumeration is received for work with Kardigan, with funds paid to C5 Research, the academic research organization of the Heart, Vascular, and Thoracic Institute of the Cleveland Clinic.
- Jones reports no relevant conflicts of interest.




Comments