LOOP and STROKESTOP: Impact of AF Screening May Hinge on Arrhythmia Burden
One trial was negative, the other positive, leading to speculation that the type of episode may be key.
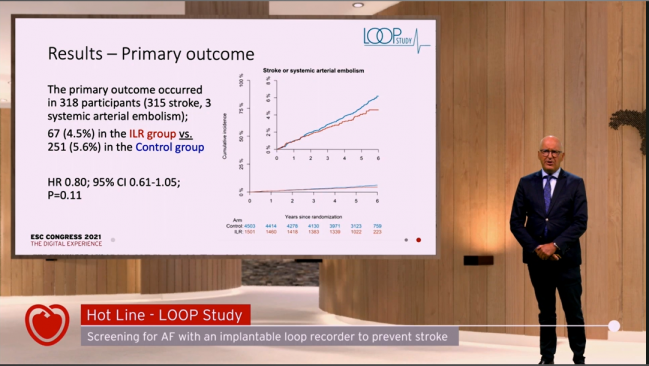
Among older people at high risk for stroke, the benefits of screening for atrial fibrillation (AF)—and then treating it when it’s detected—may be dependent on what types of episodes are being uncovered, results from the LOOP and STROKESTOP studies suggest.
In the LOOP study, looking for AF with an implantable loop recorder (ILR) and then initiating anticoagulation for any episodes lasting more than 6 minutes did not significantly reduce the risk of stroke or systemic arterial embolism compared with standard care, Jesper Svendsen, MD (Copenhagen University Hospital – Rigshospitalet, Denmark), reported during a Hot Line session at the virtual European Society of Cardiology (ESC) Congress 2021.
On the other hand, the STROKESTOP results, published in the Lancet along with LOOP but not a part of the ESC program, show that screening individuals with twice-daily ECGs for 2 weeks, then treating those with detected AF, led to a small but significant reduction in ischemic/hemorrhagic stroke, systemic embolism, bleeding resulting in hospitalization, and all-cause death (HR 0.96; 95% CI 0.92-1.00; P = 0.045).
The difference is outcomes between the two trials is likely related to the types of AF episodes detected using the respective screening approaches, according to Renate Schnabel, MD (University Heart & Vascular Center Hamburg, Germany), who told TCTMD that more-frequent and longer bouts are believed to be associated with a higher stroke risk compared with less-frequent and shorter ones. ILR monitoring picked up very short episodes in LOOP, whereas the intermittent ECG screening in STROKESTOP likely identified individuals with more clinically meaningful AF, she said. “We now need to rethink that not all atrial fibrillation is created equal.”
Indeed, the LOOP results in particular “might imply that not all atrial fibrillation is worth screening for, and not all screen-detected atrial fibrillation merits anticoagulation,” Svendsen et al write in their paper.
Taken together, Schnabel said, LOOP and STROKESTOP indicate “that screening for atrial fibrillation in individuals at risk—either by age or by enriched risk factors—may reduce stroke, systemic embolism, and mortality, and can be cost-effective, if we find the right burden of atrial fibrillation that requires anticoagulation,” Schnabel said.
The LOOP Study
The idea behind population screening for AF is that many patients with the arrhythmia don’t have symptoms, are undiagnosed, and thus are missing out on the benefits of oral anticoagulation for stroke prevention.
“European and US guidelines recommend opportunistic screening for atrial fibrillation in people aged 65 years and older using pulse-palpation or standard [ECG], whereas systematic or more-intense screening is recommended in individuals at high risk of stroke,” Svendsen et al note. “These recommendations are based on studies showing that screening is feasible and will detect more cases of atrial fibrillation, whereas the effect on stroke prevention remains unknown.”
The LOOP study, performed at four Danish centers, was designed to help address that open question. Investigators enrolled 6,004 patients ages 70 to 90 (mean age 74.7 years; 47.3% women) who had at least one of the following risk factors: hypertension, diabetes, heart failure, or prior stroke. They were randomized 1:3 to ILR monitoring—with the Reveal LINQ (Medtronic)—or standard care, which involved an annual interview with a study nurse and typical interactions with the participant’s general practitioner. Most participants (90.7%) had hypertension, and the median CHA2DS2-VASc score was 4.
As expected, through a median follow-up of 65 months, AF was more likely to be diagnosed in the ILR group (31.8% vs 12.2%; P < 0.001), in which the median duration of monitoring was 39.3 months. The rate of initiation of oral anticoagulation was higher as well (29.7% vs 13.1%; P < 0.0001).
That did not, however, translate into a significantly lower rate of stroke or systemic arterial embolism with ILR monitoring versus standard care (4.5% vs 5.6%; HR 0.80; 95% CI 0.61-1.05). A subgroup analysis suggested that screening reduced this endpoint in patients with the highest systolic blood pressure, but Svendsen stressed that the finding is only hypothesis-generating.
There were also no differences in all-cause death (11.2% vs 11.3%; HR 1.00; 95% CI 0.84-1.19), CV death (2.9% vs 3.5%; HR 0.83; 95% CI 0.59-1.16), or major bleeding (4.3% vs 3.5%; HR 1.26; 95% CI 0.95-1.69).
STROKESTOP
The STROKESTOP findings were initially presented by Emma Svennberg, MD, PhD (Karolinska University Hospital Huddinge, Stockholm, Sweden), during the virtual European Heart Rhythm Association Congress 2021 earlier this year. Residents ages 75 and 76 living in two regions of Sweden were randomized to be invited to screening, which consisted of intermittent ECG readings taken twice-daily for 2 weeks, or not.
Overall, 28,768 individuals were randomized, and about half of those invited to screening (51.3%) actually participated.
Median follow-up in STROKESTOP—6.9 years—was longer than follow-up in the LOOP study. Over that span, there was a slightly but significantly lower rate of the primary composite endpoint in the screening group (31.9% vs 33.0%; P = 0.045).
Those findings “suggest that screening for atrial fibrillation in an older population has a net beneficial effect and should be considered as part of healthcare policy,” Svennberg et al conclude.
Time to Strengthen the Guidelines?
Discussing the LOOP results after Svendsen’s presentation, Isabelle Van Gelder, MD, PhD (University Medical Center Groningen, the Netherlands), said the study helps fill in some knowledge gaps regarding which population to screen, how to do it, and the significance of a low AF burden.
She reviewed some possible explanations for the negative trial result, including the detection of short, subclinical episodes of AF; the use of a more-restrictive primary endpoint compared with STROKESTOP; the higher-than-expected rate of AF detection in the control group; and the 12% rate of early discontinuation of ILR monitoring.
“The LOOP study contributes to the evidence that short episodes of subclinical AF are not worth screening for—stroke rate is lower, no benefit of anticoagulation,” Van Gelder said. Together, she added, LOOP and STROKESTOP “suggest that EKG-detected atrial fibrillation in higher-risk patients may identify more accurately those who benefit from anticoagulation because they have clinical AF.”
But more studies of AF screening are needed, she concluded.
For Schnabel, the findings—particularly of STROKESTOP—confirm current guideline recommendations around AF screening and might support strengthening them. Guidelines state that systematic screening in individuals at least 75 years older or with additional risks factors, as done in STROKESTOP, should be considered. “This will probably get an even stronger recommendation in the next guidelines,” Schnabel predicted.
What’s important to emphasize, she added, is “that it’s not only the screening as done in these studies, but also as shown with the STROKESTOP study, there needs to be a platform for further workup, referral, [and a] decision on oral anticoagulation to really implement successful screening policies. You probably don’t need long-term intensive monitoring. Intermittent monitoring may be sufficient, but screening for atrial fibrillation in the individuals at risk should really be considered.”
Moreover, screening programs need to enhance their efforts to reach people who don’t participate, she said, noting that in STROKESTOP, the people at highest risk of adverse outcomes were those who were invited for screening but didn’t attend.
Todd Neale is the Associate News Editor for TCTMD and a Senior Medical Journalist. He got his start in journalism at …
Read Full BioSources
Svendsen JH, Diederichsen SZ, Højberg S, et al. Implantable loop recorder detection of atrial fibrillation to prevent stroke (the LOOP study): a randomised controlled trial. Lancet. 2021;Epub ahead of print.
Svennberg E, Friberg L, Frykman V, et al. Clinical outcomes in systematic screening for atrial fibrillation (STROKESTOP): a multicentre, parallel group, unmasked, randomised controlled trial. Lancet. 2021;Epub ahead of print.
Disclosures
- The LOOP study was funded by Innovation Fund Denmark, The Research Foundation for the Capital Region of Denmark, The Danish Heart Foundation, Aalborg University Talent Management Program, Arvid Nilssons Fond, Skibsreder Per Henriksen, R og Hustrus Fond, The AFFECT-EU Consortium (EU Horizon 2020), Læge Sophus Carl Emil Friis og hustru Olga Doris Friis’ Legat, and Medtronic.
- Svendsen is a member of Medtronic advisory boards and has received speaker honoraria and research grants from Medtronic.
- STROKESTOP was funded by the Stockholm County Council, the Swedish Heart & Lung Foundation, King Gustav V and Queen Victoria’s Freemasons’ Foundation, the Klebergska Foundation, the Tornspiran Foundation, the Scientific Council of Halland Region, the Southern Regional Healthcare Committee, the Swedish Stroke Fund, Carl Bennet AB, Boehringer Ingelheim, Bayer, and Bristol Myers Squibb–Pfizer.
- Svennberg reports personal fees from Bayer, Bristol Myers Squibb–Pfizer, Boehringer Ingelheim, Merck Sharp & Dohme, and Sanofi Aventis.





Comments