Lp(a) Not Associated With Ongoing Aortic Valve Calcification
The findings imply that any interventions to reduce Lp(a) would need to happen before the disease is underway, researchers say.
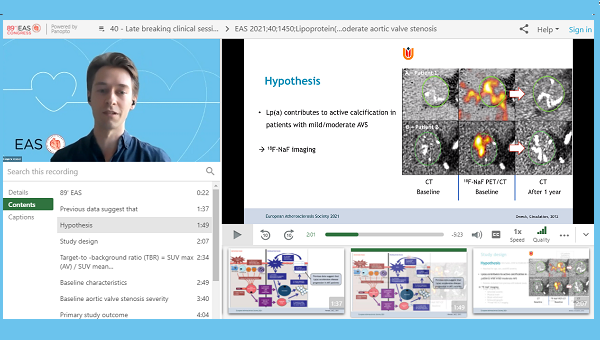
Lipoprotein(a) is not associated with active calcification of the aortic valve in patients with mild-to-moderate aortic stenosis, according to a study presented this week at the European Atherosclerosis Society Congress 2021.
In this small study of 52 patients, there was no significant difference in the extent of active aortic valve calcification as measured by 18F-NaF PET/CT imaging between those with high versus low levels of Lp(a), reported Yannick Kaiser, MD (Amsterdam University Medical Center, the Netherlands), and colleagues.
The reason, investigators suspect, is that the extent of aortic valve disease, though not severe, might have been too advanced.
“I think there’s very substantial evidence that Lp(a) is a highly likely causal risk factor for aortic valve stenosis and that by lowering it we may prevent aortic valve stenosis, but only if we lower it early enough,” said Kaiser. “I think what we saw is that we were simply too late for these people. They had too advanced aortic valve stenosis.”
To TCTMD, Kaiser explained that aortic stenosis presents almost as two distinct disease states in the initiation and propagation phases. In the initiation phase, there is damage to the valvular endothelial cells and this leads to the release of inflammatory mediators, which in turn trigger a series of reactions that result in the formation of calcium. One started, the process is a closed loop where the deposition leads to mechanical damage, apoptosis, and further valve calcification.
Once the propagation phase is in full swing, researchers believe that this process is independent from the traditional risk factors, such as Lp(a). However, there is some evidence that Lp(a) might also accelerate disease progression during the propagation phase in patients with aortic stenosis, as shown in a 2019 post hoc analysis.
No Difference in Active Calcification
With that in mind, the researchers wanted to determine if Lp(a) contributed to active calcification in patients with mild-to-moderate aortic valve stenosis. The study included 26 patients with high Lp(a) levels (mean 79 mg/dL) and 26 with low Lp(a) levels (mean 5 mg/dL). Patients were well matched by baseline aortic stenosis severity at baseline—the mean aortic valve gradient was 17.9 mm Hg in both groups—although patients with low Lp(a) levels had higher systolic blood pressure and LDL-cholesterol levels.
The researchers used 18F-NaF PET/CT imaging to assess the target-to-background ratio (TBR), which measures the uptake of 18F-NaF corrected for uptake in the right atrium. Aortic valve TBRmax, the study’s primary endpoint, has been used in previous studies because it has the most robust relationship with subsequent calcification, said Kaiser. However, “completely contrary to our hypothesis, we observed no difference in 18F-NaF uptake in the valve,” he said.
In a linear regression analysis, none of the traditional risk factors—age, male sex, high Lp(a), elevated systolic blood pressure, or elevated LDL cholesterol—were associated with active calcification. In that analysis, only the aortic valve calcification score on CT (per 1,000-AU increase) was associated with aortic valve calcification.
“Even though we have a relatively mild disease stage in this study, already the calcium score was the only thing that determined progression,” said Kaiser.
In Europe, the clinical guidelines recommend checking Lp(a) for CVD risk stratification in individuals at least once during the patient’s lifetime, although the recommendation is relatively weak. At present, Lp(a) is mostly used as a risk-enhancing factor to help further clarify a patient’s CVD profile. The Lp(a)HORIZON study is ongoing, with investigators testing whether the Lp(a)-lowering antisense oligonucleotide previously known as TQJ230 can reduce the risk of MACE when added to guideline-recommended therapy.
Kaiser said that the relationship between Lp(a) and aortic valve calcification raises the possibility that Lp(a)-lowering might be the first medical intervention for aortic valve stenosis, but that it would need to be tested in randomized clinical trials. Based on the present study, however, the data would suggest the need to assess Lp(a) levels—and potentially intervene—well before the calcification process has started, he said.
Michael O’Riordan is the Managing Editor for TCTMD. He completed his undergraduate degrees at Queen’s University in Kingston, ON, and…
Read Full BioSources
Kaiser Y, Nurmohamed NS, Kroon J, et al. Lipoprotein(a) is not associated with active calcification assessed with 18F-NaF PET/CT in patients with mild to moderate aortic valve stenosis. Presented at: EAS 2021. June 2, 2021.
Disclosures
- Kaiser reports no conflicts of interest.



Comments