TAVR-MET: Early Signs Point to Less Postprocedural Valve Dysfunction With Tirzepatide
Patients with obesity randomized to the drug had less subclinical leaflet thrombosis and paravalvular leak than controls.
WASHINGTON, DC—Patients with obesity who are on tirzepatide (Zepbound; Eli Lilly) prior to undergoing TAVI may have a lower risk of developing subclinical leaflet thrombosis and paravalvular leak (PVL), a small, randomized trial suggests.
Compared with those who were not taking the weight-loss medication before and after the procedure, those who were had a 61% reduction in the relative risk of grade 1 to 4 hypoattenuated leaflet thickening (HALT) at 12 months (8.4% vs. 21.6%; P = 0.002). The risk of having at least mild PVL, a secondary endpoint, was 10.7% in those on tirzepatide compared with 25.3% in the control arm (P = 0.006), corresponding to a 58% reduction.
“The TAVR-MET trial provides the first evidence that metabolic modulation can improve bioprosthetic valve healing and hemodynamics in obesity-associated aortic stenosis,” said A.M. Thirugnanam, MD (Ipcard Cardiac Care Center, Hyderabad, India), in a presentation last week at CRT 2026.
He added that while the mechanism behind the observation is unclear, it may be related to attenuation of proinflammatory markers and/or weight loss. Tirzepatide is a dual agonist of the glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) receptors.
Thirugnanam noted that obesity “drives a proinflammatory, prothrombotic state” and its prevalence is more than 40% in contemporary TAVI practice. Additionally, he said HALT has been shown to occur in 15% to 25% of patients and has been linked to serious adverse outcomes.
Session co-moderator Ron Waksman, MD (MedStar Washington Hospital Center, Washington, DC), noted that while GIP/GLP-1 therapy has been shown to help patients with obesity and heart failure, he was surprised to see that it also may have an impact on HALT. He called on US investigators to corroborate the trial’s findings.
“It's a relatively impressive result in a relatively small number of patients,” added Howard C. Herrmann, MD (Perelman School of Medicine at the University of Pennsylvania, Philadelphia), a panelist in the late-breaking trial session. “It begs for confirmation and a bigger trial, and it raises the question of whether this is a pleotropic effect of the drug, versus only a weight loss effect.”
TAVR-MET trial
Thirugnanam and colleagues randomized 260 patients (mean age 73 years; 47% women) with a body mass index of 30 kg/m2 or greater to tirzepatide once weekly starting 4 weeks before the procedure and continuing for 12 months, or to standard care. Tirzepatide was escalated at the recommended intervals starting at a dose of 2.5 mg per week for 4 weeks, followed by 5 mg per week for 4 weeks and then 10 mg per week for the duration of the study period. Patients in both groups received standard dual antiplatelet therapy consisting of aspirin 75 mg and clopidogrel 75 mg for 3 months. The mean weight loss was 9.2 kg in the tirzepatide group and 1.6 kg in controls (P < 0.001).
In addition to the main results, the progression of subclinical leaflet thrombosis was significantly lower in the tirzepatide-treated patients (3.1% vs 11.5% in controls; P = 0.01).
An analysis of metabolic and inflammatory markers showed a reduction in C-reactive protein levels of 38% with tirzepatide versus 10% in controls (P < 0.001). Tirzepatide also was associated with better suppression of interleukin-6 (P < 0.001) and tumor necrosis factor (TNF) alpha (P = 0.003).
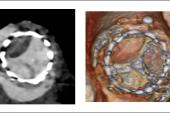
The HALT distribution grades at 12 months, as confirmed on transesophageal echocardiography, all favored tirzepatide in comparison with controls, as did mean gradient and effective orifice area.
It begs for confirmation and a bigger trial, and it raises the question of whether this is a pleotropic effect of the drug, versus only a weight loss effect. Howard C. Herrmann
The incidence of major adverse valve events (MAVE), a composite of death, stroke, valve reintervention, and structural valve deterioration, was higher in controls than in the tirzepatide arm, but the difference did not reach statistical significance, with Thirugnanam noting that the trial was underpowered for the composite. Among other clinical endpoints, there was a trend toward less stroke and TIA in the tirzepatide group, as well as lower risk of hospitalization for HF.
Panelist Chad Kliger, MD (Lenox Hill Heart and Lung Institute, New York, NY), called the data provocative.
“We've known about HALT for the last 12 years, but really haven't moved forward much in this space,” he noted, adding that he looks forward to the dataset being peer reviewed and published to better understand the potential impact of GLP-1 medications on valve hemodynamics.
“Will I prescribe [tirzepatide] to my patients? Personally, no. But I think in the future it may be reasonable to do so and . . . to make this a potential treatment pathway,” Kliger added.
L.A. McKeown is a Senior Medical Journalist for TCTMD, the Section Editor of CV Team Forum, and Senior Medical…
Read Full BioSources
Thirugnanam AM. TAVR-MET trial: tirzepatide as a novel cardioprotective strategy to reduce subclinical leaflet thrombosis after TAVR. Presented at: CRT 2026. March 9, 2026. Washington, DC.
Disclosures
- Thirugnanam reports no relevant conflicts of interest.



Comments