Calcified Nodules Signal Worst Outcome Among OCT-Defined ACS Causes
The TACTICS results confirm prior studies, Ziad Ali says. “Whether OCT could help us . . . alter those outcomes remains to be seen.”
BOSTON, MA—Most cases of acute coronary syndrome are caused by plaque rupture, but those tied to calcified nodules carry the greatest risk of MACE at 1 year, according to the largest prospective optical coherence tomography (OCT) study to address these issues to date.
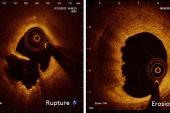
In the TACTICS registry of patients who underwent OCT-guided primary PCI, plaque rupture was the underlying ACS cause in 59% of patients, plaque erosion in 26%, and a calcified nodule in 4%, with a mix of other causes making up the rest, Toshiro Shinke, MD, PhD (Showa University Hospital, Tokyo, Japan), reported here at TCT 2022.
Those with calcified nodules fared the worst through 1 year, with a 32.1% rate of MACE (CV death, MI, heart failure, or ischemia-driven revascularization). That compares with rates of 6.2% and 12.4% among patients with plaque erosions or ruptures, respectively.
The registry also showed that OCT led to a change in PCI strategy in most patients (58%). Future studies “to evaluate the possibility of OCT-guided optimization based on ACS underlying causes is warranted,” Shinke said at a press conference.
Commenting for TCTMD, Ziad Ali, MD, DPhil (St. Francis Hospital & Heart Center, Roslyn, NY), praised the scientific rigor of the study, which he said confirmed prior investigations in terms of the procedural impact of OCT, as well as the distribution of the underlying causes of ACS and associated clinical outcomes. “There’s a lot of new data to suggest that despite good acute procedural success, these calcified nodule patients are doing poorly at 1 year,” he said, noting that there could be some confounding in the finding since these patients tend to be older and have poorer kidney function.
As for whether OCT guidance can improve clinical outcomes, Ali said that remains to seen. He pointed out that the ILUMIEN IV trial comparing OCT-guided versus angiography-guided PCI in high-risk patients—for which he serves as the global principal investigator—is set to answer that question. The trial recently completed enrollment with 2,690 patients, with initial results expected next year.
TACTICS Registry
Prior retrospective studies have suggested that OCT enables the diagnosis of the underlying causes of ACS, but there were still uncertainties about their prevalence and their relationships with long-term clinical outcomes, Shinke said.
The prospective TACTICS registry, which involved 22 hospitals in Japan, was set up to address those questions. The analysis included 702 patients who were diagnosed with ACS within 24 hours of symptom onset and who underwent OCT-guided primary PCI. Most patients (63%) had STEMI, 28% NSTEMI, and 9% unstable angina.
For PCI, recanalization was achieved before OCT was used to define the underlying cause, to measure vessel size and guide stent sizing, and to optimize the procedural result with a goal of TIMI 3 flow with either a minimum stent/lumen area ≥ 4.5 mm2 or a percent stent expansion ≥ 70%, with no major dissection, large malapposition, extensive protrusion, or thrombus.
Overall, 97% of patients achieved TIMI 3 flow, and 85% obtained a minimum stent/lumen area ≥ 4.5 mm2 or a percent stent expansion ≥ 70% at the end of the procedure, with no differences based on the underlying cause of ACS.
Despite similar procedural results, however, outcomes differed by underlying cause. After adjustment, risk of 1-year MACE was significantly higher in patients with calcified nodules (HR 4.52; 95% CI 1.54-13.27) and nonsignificantly higher in those with plaque rupture (HR 1.88; 95% CI 0.96-3.67) compared with patients with erosions.
OCT findings influenced the PCI strategy in most patients. Most commonly it led to additional postdilatation (54.6% of cases) and changes to stent length (16.0%) and size (15.7%). In smaller proportions of patients, the imaging results resulted in a stentless procedure, additional lesion preparation, or the use of more stents.
Importance of Assessing Lesion Morphology
Shinke acknowledged some limitations of the study, including possible selection bias stemming from the focus only on ACS patients who underwent OCT-guided primary PCI; the lack of histological confirmation of the OCT-defined underlying causes of ACS; and the lack of standardization of the OCT-guided strategy according to the plaque morphology.
Even so, Gary Mintz, MD (Cardiovascular Research Foundation, New York, NY), highlighted why these types of studies into what’s causing ACS are important. Prior data have suggested that some plaque erosions can be treated with medical therapy and not stenting, whereas multiple trials have indicated that the best way to treat plaque ruptures is with stent implantation, he noted. And there is this third group, those with calcified nodules, who have been shown to have the worst outcomes.
“We know that when these recur, or when these cause events—particularly the erupted calcified nodules, which is what these are—they are associated with re-intrusion of the calcium into the stent, whether stent thrombosis or restenosis,” Mintz said. “And it’s not clear whether it’s because of stent fracture at the time of stent implantation or a biologic process of reaccumulation of the erupted nodule inside the stent.
“But clearly the morphologic distinction among primary PCI lesions is related to long-term outcome,” he continued. “And that’s why these kinds of studies are so important.”
Todd Neale is the Associate News Editor for TCTMD and a Senior Medical Journalist. He got his start in journalism at …
Read Full BioSources
Shinke T. Impact of underlying causes of acute coronary syndrome on 1-year outcomes after percutaneous coronary intervention. Presented at: TCT 2022. September 19, 2022. Boston, MA.
Disclosures
- Shinke reports grant/research support from Abbott Medical Japan and consulting fees/honoraria from Abbott Medical Japan, Bayer, Boston Scientific, Bristol Myers Squibb, and Daiichi Sankyo.
- Ali reports receiving institutional grant support from Abbott and being the global principal investigator of ILUMIEN IV, which is sponsored by Abbott.
- Mintz reports fees related to consulting or speaking from Abiomed, Boston Scientific, and Medtronic.



Comments