IVUS-Guided PCI Tied to Better Long-term Outcomes After Acute MI
“IVUS should certainly be used more frequently based on the accumulated evidence,” Arnold Seto says.
Another study, this one a large registry analysis from Korea with long-term follow-up, has provided support for use of IVUS-guided PCI in patients with acute myocardial infarction.
After 4 years, patients whose drug-eluting stent implantation was guided by IVUS versus angiography had lower risks of MACE, target lesion revascularization, and both cardiovascular and all-cause death, according to findings from the COREA-AMI registry published online this week ahead of print in JACC: Cardiovascular Interventions.
That bolsters prior research, including the IVUS-XPL and ULTIMATE randomized trials, demonstrating the benefits of using IVUS to ensure optimal stent expansion in broader groups of patients undergoing PCI, mostly due to reductions in TLR and TVR. These data extend those results by adding a potential long-term reduction in mortality.
“The use of IVUS in PCI should be considered for patients with acute MI,” conclude the researchers, led by Ik Jun Choi, MD, PhD, and Sungmin Lim, MD, PhD (both Catholic University of Korea, Seoul).
Across all types of patients undergoing PCI, use of IVUS varies widely around the world thanks to differences in reimbursement, Arnold Seto, MD (University of California, Irvine, and VA Long Beach Healthcare System), commented to TCTMD. The modality is deployed in less than 5% of PCIs in Europe, where there is no reimbursement; in 9% to 15% of cases in the US, where there is partial reimbursement; and in up to 90% of procedures in Korea and Japan, where there is a separate payment to cover its use.
“If that’s not a natural experiment in use of IVUS being due to reimbursement, I don’t know what is,” Seto said, adding that it is likely used less often in patients with acute MI than in those undergoing elective procedures.
But based on the accumulated evidence from registry studies like this one, randomized trials, and meta-analyses, Seto said, “I think that IVUS should certainly be used more frequently.”
COREA-AMI
Though international practice guidelines have endorsed the use of IVUS (as well as optical coherence tomography) to optimize stent implantation, its impact on long-term clinical outcomes has remained unclear.
To explore that issue, Choi, Lim, and colleagues turned to the COREA-AMI registry, examining data on 9,846 Korean patients with acute MI (mean age 63.3 years; 72.1% men) who underwent PCI with DES between January 2004 and August 2014. Based on the discretion of the treating physician, IVUS was used to guide the procedure in 20.6% of cases, with angiographic guidance employed for the rest.
Procedural complications did not differ between the IVUS- and angiography-guided groups, but longer-term outcomes evaluated over 4 years of follow-up favored IVUS guidance. The primary outcome of MACE (CV death, MI, and TLR) occurred in 15.3% of patients in the IVUS group and 19.0% of those in the angiography group (HR 0.78; 95% CI 0.69-0.88), with the difference remaining significant after multivariable regression and propensity-score matching.
A landmark analysis showed that IVUS guidance was associated with a lower MACE risk both in the first year (adjusted HR 0.81; 95% CI 0.68-0.96) and from 1 to 4 years (adjusted HR 0.80; 95% CI 0.66-0.960).
The IVUS group also had lower rates of CV death (10.8% vs 13.3%; HR 0.79; 95% CI 0.69-0.91), TLR (3.4% vs 4.4%; HR 0.74; 95% CI 0.57-0.96), and all-cause death (12.9% vs 16.9%; HR 0.75; 95% CI 0.64-0.85). In a landmark analysis, however, the risk of CV death differed between groups only in the first year of follow-up, with the difference in TLR only seen from 1 to 4 years.
As for repeat infarction, IVUS guidance was associated with a lower rate of culprit MI (1.6% vs 2.4%; HR 0.65; 95% CI 0.45-0.94), but not nonculprit MI. Rates of stent thrombosis were similar in the two groups.
How Does IVUS Help?
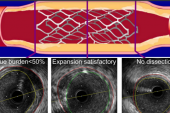
Seto said the exact mechanisms underlying the beneficial effects of IVUS guidance are not clear. “However, we know that IVUS increases the minimal stent area. And at least in this study but also in some other studies, it reduces target lesion revascularization, which may be due to restenosis because of underexpansion of the stent,” he explained, “or it could be due to stent thrombosis, also due to underexpansion of the stent.”
Other possibilities are related to the detection of calcium with IVUS—which allows for better lesion preparation to avoid underexpansion—or more-complete coverage of lesions. With angiography, many operators will stent only the tightest lesion, Seto said. In contrast, with IVUS “you’ll often see more diffuse disease and long disease, and at least in this study, IVUS use was associated with more and longer stents, and that might drive people to have better lesion coverage and not have as much geographic miss.”
Indeed, the investigators agree, the fact that IVUS-guided PCI was associated with more stents implanted, a larger mean stent diameter, and a longer total stent length “could influence full lesion coverage and stent optimization, which might lower the incidence of poor clinical outcomes.”
Still, they say, “large, randomized trials, registries, national data, or systematic reviews and meta-analyses are necessary to confirm the short-term and long-term impacts of IVUS-guided PCI on cardiovascular death, MI, or TLR.”
Seto said that there is now enough data to support IVUS, but that it will take a combination of further studies and political will to increase reimbursement for its use by government payers like the US Centers for Medicare & Medicaid Services. At least in the US, physicians will have to go to bodies like the American Medical Association (AMA) to argue for more reimbursement, understanding that that might come at the expense of other things done by interventional cardiologists. “That’s that zero-sum game that happens at the AMA and often is an impediment to having high-quality, proven, and potentially high-value services like [fractional flow reserve] and IVUS from being widely adopted,” Seto said.
In an accompanying editorial, Yasuhiro Honda, MD (Stanford University School of Medicine, CA), notes the limitations of observational data and highlights the questions around the mechanisms of benefit with IVUS guidance, but argues against the need for much more randomized evidence.
“As the body of evidence in this arena has nearly matured with consistent results supporting the clinical benefit and cost-effectiveness of IVUS-guided PCI, further designing mega-scale randomized controlled trials to merely confirm the advantage of IVUS guidance for each complex lesion/high-risk patient subset would not be warranted anymore,” he says. “Future efforts should rather be directed towards the establishment of standardized workflow to maximize the benefit of image guidance in various lesion/patient subsets.
“In this context, rapidly evolving digital health technologies and emerging application of artificial intelligence may help in developing the algorithm and system to assist operators in catheterization laboratories,” he continues. “The incremental roles of commercially available, multimodality hybrid imaging catheters, such as NIRS-IVUS and OCT-IVUS, may also be of great interest, particularly in the field of primary PCI.”
Todd Neale is the Associate News Editor for TCTMD and a Senior Medical Journalist. He got his start in journalism at …
Read Full BioSources
Choi IJ, Lim S, Choo EH, et al. Impact of intravascular ultrasound on long-term clinical outcomes in patients with acute myocardial infarction. J Am Coll Cardiol Intv. 2021;Epub ahead of print.
Honda Y. Intravascular imaging to guide PCI for acute myocardial infarction: shifting from “whether” to “how”. J Am Coll Cardiol Intv. 2021;Epub ahead of print.
Disclosures
- Choi, Lim, Chang, and Honda report no relevant conflicts of interest.
- Seto reports research grants from Acist and Philips.


Comments