SURTAVI: Self-Expanding TAVI Valve Holds Up vs SAVR Through 5 Years
(UPDATED) After 2 years, clinical outcomes were similar, hemodynamics favored TAVI, and paravalvular leak favored surgery.

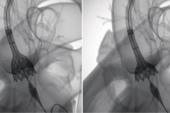
ORLANDO, FL—(UPDATED) Through 5 years, TAVI with a self-expanding valve provided clinical outcomes similar to those seen with surgery in an intermediate-risk population, according to continued follow-up of the SURTAVI trial.
As seen at 2 years, the rate of all-cause mortality or disabling stroke was no different between the TAVI and SAVR arms at 5 years (31.3% vs 30.8%; HR 1.02; 95% CI 0.85-1.22), Nicolas Van Mieghem, MD, PhD (Thoraxcenter, Erasmus University Medical Center, Rotterdam, the Netherlands), reported here at TCT 2021.
Hemodynamics remained superior with TAVI through 5 years, whereas surgery continued to hold the advantage in terms of paravalvular leak (PVL). Though rates of reintervention and permanent pacemaker implantation were higher in the TAVI arm, there were no differences in these outcomes—or any other key endpoints—between 2 and 5 years.
Van Mieghem noted, too, that there were low rates of valve thrombosis and endocarditis in both groups.
“Long-term outcome data from the SURTAVI randomized trial comparing early-generation TAVR to open-heart surgery are similar and encouraging for TAVR for younger, healthier patients with aortic stenosis,” he concluded.
Commenting for TCTMD, Milan Milojevic, MD, PhD (Dedinje Cardiovascular Institute, Belgrade, Serbia), chair of the practice guidelines committee of the European Association for Cardio-Thoracic Surgery, said these longer-term data “have bolstered support for TAVI as a complementary treatment procedure in elderly patients at intermediate-to-high surgical risk.”
In addition, he said, they are consistent with the recently updated European valvular heart disease guidelines, which incorporated 5-year data from the PARTNER 2A trial showing similar outcomes with TAVI using a balloon-expandable valve and with SAVR in intermediate-risk patients. Milojevic served on the task force for the guidelines.
He stressed, however, that SAVR should not be discarded as an option in this group, noting that the much-greater cost of TAVI must be considered, especially in middle-income countries. He also provided notes of caution related to the population enrolled in the trial, which had an average age of 80 and a life expectancy of less than 10 years, and to the higher rates of PVL and pacemaker implantation in the TAVI arm. Those issues “should be important considerations before TAVI . . . moves into the younger population of patients,” Milojevic said. It’s crucial to better understand their impact, which can only be done with longer-term follow-up in the trials of low-risk patients who do not have as high a risk of competitive noncardiovascular events as the population enrolled in SURTAVI, he added.
“It is important for both physicians and patients to indicate or undergo the treatment according to the best practice data, and we should not go ahead of the data,” he said.
During a panel discussion following Van Mieghem’s presentation, Patrick O’Gara, MD (Brigham and Women’s Hospital, Boston, MA), was similarly circumspect when looking at the findings, which he called “reassuring with respect to durability among patients whose average age is 80 who receive this particular device.”
But he raised a concern about “indication creep,” stating that physicians need to be cautious in translating the outcomes to younger patients at lower risk. “It’s not really fair for us to say 60-year-old patients will fare equally well or that the tradeoff between a pacemaker implant and the need for a repeat procedure is perceived the same in folks who are 15 years younger.”
For 80-year-olds weighing the choice between TAVI and SAVR, “they’d be more than happy to put up with a pacemaker compared against a median sternotomy. I think that’s the tradeoff our patients have already made,” O’Gara said. “But let’s be careful about how we define younger patients and lower risk.”
The SURTAVI Trial
Across 87 centers in the United States, Europe, and Canada, SURTAVI randomized 1,746 patients with intermediate surgical risk (mean STS PROM score 4.5%) to TAVI with a self-expanding supra-annular valve or SAVR, after stratification for the need for coronary revascularization. In the TAVI arm, most patients (84%) were treated with the first-generation CoreValve, with the rest receiving the Evolut R (both Medtronic).
Van Mieghem reported the 5-year echocardiographic and clinical outcomes. Regarding hemodynamics, TAVI provided larger effective orifice areas and lower mean gradients compared with surgery at each follow-up time point through 5 years (P < 0.001 for all).
But surgery topped TAVI when it came to the amount of paravalvular leak, a difference seen early and then maintained through 5 years. At that point, patients in the SAVR arm had lower rates of mild PVL (2.7% vs 27.1%) and moderate PVL (0.7% vs 3.0%), with no severe cases in either group. Similar findings were seen for total aortic regurgitation.
The lack of difference in all-cause mortality or disabling stroke between trial arms was seen over the course of the entire 5-year follow-up, and a landmark analysis showed that the finding was consistent both within the first 2 years and from 2 to 5 years.
As previously reported at 2 years, TAVI was associated with a higher rates of reintervention (3.5% vs 1.9%; P = 0.02) and permanent pacemaker implantation (30.9% vs 9.8%; P <0.001) at 5 years. Van Mieghem noted, however, that from 2 to 5 years, there were no between-group differences for these or other outcomes, and also said that pacemaker implantation was not associated with mortality at 5 years.
The rapid improvements seen previously in quality of life according to the Kansas City Cardiomyopathy Questionnaire and in NYHA functional class in both the TAVI and SAVR arms were sustained through 5 years, with no between-group differences.
Making the Choice
Commenting on the results during a panel discussion at a media briefing, surgeon Keith Allen, MD (Saint Luke's Mid America Heart Institute, Kansas City, MO), said that through 5 years, SURTAVI “continues to show that if you’re not going to get a mechanical valve, TAVR is . . . a really great option, and I think it should be first in the armamentarium.”
As for whether the results should strengthen TAVI’s place in practice guidelines, surgeon Michael Borger, MD, PhD (Leipzig Heart Center, Germany), said, “I would ask: how could it possibly strengthen the position? At least in Germany, intermediate-risk patients are all treated with TAVR.” In patients with ages and STS scores similar to those in the SURTAVI trial there is very little surgery being performed currently, he added.
Indeed, the results are “very reassuring” for TAVI, Van Mieghem said, noting that “the added value of this less-invasive therapy is the faster recovery of these patients.” The choice between surgery and TAVI will always be made using a shared decision-making process, he said, but in the elderly population, “I think it will be quite reasonable to say that most patients would feel—and should feel—comfortable when they opt for a TAVR procedure for symptomatic severe aortic stenosis.”
When discussing TAVI in patients who are younger, lower-risk, or both, experts highlighted the need to consider how treatment choices will affect decisions several years or even decades down the road. Durability is a key consideration, Allen indicated. “How do you manage these patients when they’re 55, 58, 60? Because transcatheter valves will fail in those patients, just like surgical tissue valves fail. There’s no hint that TAVR valves are going to last longer.”
Luca Testa, MD, PhD (San Raffaele Hospital, Milan, Italy), agreed, saying that when heart teams meet to discuss, they must consider “that that is just the beginning, not the end, of the valve journey.”
Todd Neale is the Associate News Editor for TCTMD and a Senior Medical Journalist. He got his start in journalism at …
Read Full BioSources
Van Mieghem NM. 5-year clinician and echocardiographic outcomes from the randomized SURTAVI trial. Presented at: TCT 2021. Orlando, FL. November 5, 2021.
Disclosures
- SURTAVI was funded by Medtronic.
- Van Mieghem reports research grants from Abbott Vascular, Boston Scientific, Edwards Lifesciences, Medtronic, Daiichi Sankyo, Abiomed, PulseCath BV, and Pie Medical.
- O’Gara reports serving on executive committees for trials run by Edwards Lifesciences and Medtronic.
- Allen reports grant support/research contracts (institutional) from Edwards Lifesciences, Medtronic, and Abbott Vascular; consultant fees/honoraria/speakers bureau fees (institutional) from Edwards Lifesciences, Medtronic, and Abbott Vascular; and consultant fees/honoraria/speakers bureau fees (personal) from Silk Road.
- Borger reports consultant fees/honoraria/speakers bureau fees (institutional) from Edwards Lifesciences, Medtronic, Abbott Vascular, and CryoLife.
- Milojevic and Testa report no relevant conflicts of interest.



Comments