COVID-19: TCTMD’s Daily Dispatch for March
We’re curating a list of COVID-19 research and other useful content, and updating it daily.

TCTMD reporter Todd Neale is keeping up on breaking news and peer-reviewed research related to COVID-19 and will update daily. If you have something to share, tell us.
March 31, 2020
A global ACS COVID-19 registry has been launched.
Researchers address the hot topic of renin-angiotensin-aldosterone system inhibitor use in patients with COVID-19 in a special report in the New England Journal of Medicine. They conclude: “Until further data are available, we think that RAAS inhibitors should be continued in patients in otherwise stable condition who are at risk for, being evaluated for, or with Covid-19.”
 The European Society of Cardiology has cancelled EuroHeartCare 2020, which was scheduled for June 4 to 6 in Seville, Spain. The society has now cancelled four of its meetings in the first half of the year. Frontiers in CardioVascular Biomedicine, held every other year, has been postponed until April 2021.
The European Society of Cardiology has cancelled EuroHeartCare 2020, which was scheduled for June 4 to 6 in Seville, Spain. The society has now cancelled four of its meetings in the first half of the year. Frontiers in CardioVascular Biomedicine, held every other year, has been postponed until April 2021.
The American Heart Association (AHA), the American College of Cardiology (ACC), the Society for Cardiovascular Angiography and Interventions (SCAI), and several other professional organizations have issued an urgent call for the federal government to address critical shortages of ventilators, test kits, and personal protective equipment (PPE).
The AHA has also provided an overview of what people with high blood pressure need to know about COVID-19.
Two new papers in the Annals of Internal Medicine discuss (1) whether the rush to discover treatments for COVID-19 may “lead to relaxed standards of data generation and interpretation, which may have undesirable downstream effects,” using the recent publication of a study of hydroxychloroquine as an example, and (2) how the SARS-CoV-2 coronavirus has been found in sputum and fecal samples after pharyngeal specimens were no longer positive.
The US Food and Drug Administration provided an update as to how it’s expediting the review of diagnostic tests for COVID-19, saying that it “has been providing unprecedented flexibility to labs and manufacturers to develop and offer COVID-19 tests across the US.”
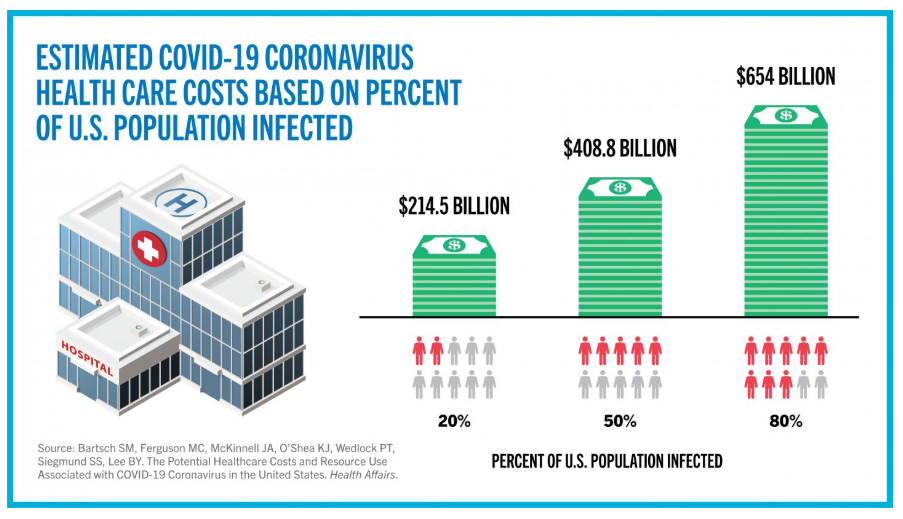
 A paper in the Lancet Infectious Diseases presents estimates of the proportion of people with COVID-19 who either require hospitalization or die. Both percentages increase sharply with age. “Our estimate of the proportion of infected individuals requiring hospitalization, when combined with likely infection attack rates (around 50–80%), show that even the most advanced healthcare systems are likely to be overwhelmed,” the investigators write.
A paper in the Lancet Infectious Diseases presents estimates of the proportion of people with COVID-19 who either require hospitalization or die. Both percentages increase sharply with age. “Our estimate of the proportion of infected individuals requiring hospitalization, when combined with likely infection attack rates (around 50–80%), show that even the most advanced healthcare systems are likely to be overwhelmed,” the investigators write.
Palliative care physicians have created a COVID-19 palliative care plan, published in CMAJ. “The SARS-CoV-2 pandemic has been tragic for many people worldwide,” the authors say. “Failing to provide Canadians with effective palliative care would compound that tragedy.”
The European Medicines Agency (EMA) issued an update Tuesday on treatments and vaccines for COVID-19 that are under development. The main takeaway? “No medicine has yet demonstrated efficacy in treating COVID-19.”
March 30, 2020
In the same TCTMD series, and originally broadcast live on Twitter, Juan Granada, MD, Azeem Latib, MD, Mamas Mamas, BMBCh, DPhil, and Federico Pappalardo, MD, discuss hospital preparedness and management protocols as well as pulmonary care for the non-pulmonary specialist in this special edition webcast on COVID-19.

Over the weekend, President Donald Trump extended US federal distancing guidelines until April 30, after one of his advisors, Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Disease, had earlier presented estimates that 200,000 Americans could die from COVID-19. Read more here in the New York Times.
The potential cardiovascular effects of coronaviruses are detailed in a review in JAMA Cardiology.
Edwards Lifesciences has paused enrollment in pivotal mitral and tricuspid trials due to the pandemic.
New guidance from the Society of Cardiovascular Computed Tomography addresses the use of cardiac CT in the context of COVID-19. It has been endorsed by the American College of Cardiology and included on the college’s COVID-19 hub.

The FDA issued a warning that chloroquine phosphate intended to treat disease in aquarium fish should not be used as a treatment for COVID-19 in humans after at least one person died doing so. The agency also provided an update on what it’s doing to minimize supply interruptions of food and medical products during the pandemic and released guidance for industry regarding how to notify the FDA of a permanent discontinuance or interruption in manufacturing.
Abbott announced that the FDA has issued an emergency use authorization for a COVID-19 test—run on the ID NOW platform—that can provide positive results in as little as 5 minutes and negative results in 13 minutes.
A 27-year-old astrophysicist and research fellow at a Melbourne university was admitted to the hospital after getting four powerful neodymium magnets stuck up his nose when trying to invent a necklace device that would sound an alarm warning people they are about to touch their face. His idea failed.
March 27, 2020
TCTMD reporters look at detailed cath-lab advice in the COVID-19 era, new data on myocardial injury, and the interaction of ACE inhibitors and ARBs with SARS-CoV-2. A feature story explores the confusing mix of policies guiding the use of masks across the United States.
The European Association of Cardiovascular Imaging is mourning the death of longtime member and EuroEcho meeting co-chair, Maurizio Galderisi, MD (Federico II University of Naples, Italy), age 65, from COVID-19. The Guardian newspaper reports that 41 health professionals in Italy have died from the disease, likely contracting the virus early in the outbreak, when protective equipment was lacking.
— SIC Società Italiana di Cardiologia (@SIC_CARDIOLOGIA) March 27, 2020
The US Food and Drug Administration has released guidance to clarify the regulatory landscape around face masks and respirators with the aim of expanding the availability of general-use face masks for the public and filtering facepiece respirators, including N95 respirators, for healthcare workers.
Today marks the debut of a special Rox Heart Radio series focused on COVID-19. In our first episode of From the Front, Roxana Mehran, MD, speaks with Giulio Stefanini, MD, PhD, and Gennaro Giustino, MD.
New research out of China, published in JAMA Cardiology, confirms other recent reports of higher mortality in patients with established CVD, further noting that of the nearly 28% of patients with myocardial injury indicated by troponin T (TnT) elevation, mortality was markedly higher than in patients with no TnT rise (59.6% vs 8.9%). Of note, say the authors, use of ACEI/ARBs did not appear to significantly influence survival in this series.

COVID-19 needs a Manhattan Project, argues Seth Berkley, CEO of Gavi, the Vaccine Alliance, in Science. “If ever there was a case for a coordinated global vaccine development effort using a ‘big science’ approach, it is now,” he writes.
 Two research letters in JAMA address the risks of maternal transmission of the virus. The first describes the possible ‘vertical’ transmission of COVID-19 from an infected mother to her newborn. The second research letter, however, reports that six infants from mothers with confirmed COVID-19 did not have SARS-CoV-19 detected in their serum or by throat swab using reverse transcription-polymerase chain reaction (RT-PCR), although virus-specific antibodies were detected in neonatal blood samples.
Two research letters in JAMA address the risks of maternal transmission of the virus. The first describes the possible ‘vertical’ transmission of COVID-19 from an infected mother to her newborn. The second research letter, however, reports that six infants from mothers with confirmed COVID-19 did not have SARS-CoV-19 detected in their serum or by throat swab using reverse transcription-polymerase chain reaction (RT-PCR), although virus-specific antibodies were detected in neonatal blood samples.
Alberta researchers are proposing a “4-level” strategy to help triage extracorporeal life support, given limited access in many jurisdictions.
The National Library of Medicine is expanding access to scientific papers on COVID-19 through PubMed Central, following the lead of medical journals and newspapers that have ungated access to COVID-19 research and coverage. TCTMD is also providing free access to all content during this period, including live cases, videos, and more.
A study in the Lancet Public Health examines the impact of control strategies to reduce social mixing on the outcome of the COVID-19 outbreak in Wuhan, China. Acknowledging uncertainties in the data, the researchers conclude that “restrictions on activities in Wuhan, if maintained until April, would probably help to delay the epidemic peak. Our projections suggest that premature and sudden lifting of interventions could lead to an earlier secondary peak, which could be flattened by relaxing the interventions gradually.”
With reporting from Shelley Wood.
March 26, 2020
TCTMD’s L.A. McKeown reports on how the experience of Chinese physicians may help others as the pandemic explodes in the United States and around the world.
New York University’s Grossman School of Medicine is planning on allowing select medical students to graduate early in order to help in the COVID-19 fight.
Two new papers have been published in Circulation. The first discusses how to apply the lessons of influenza to the novel coronavirus behind COVID-19 (SARS-CoV-2). In the second, Italian researchers use their experience to detail critical organizational issues for cardiologists during the pandemic.
Brigham and Women’s Hospital’s COVID-19 critical care clinical guidelines, which are being updated daily, can be found here.
A perspective in the New England Journal of Medicine delves into how to prepare the health system for the pandemic, and at the same time asks why we have not taken the right steps after prior crises to prepare for future ones. “Because we fail to learn the lessons and dedicate the funding and planning efforts required. Because doing so is not prioritized by regulators, payers, or most hospital leaders. Because the need is not understood by the public. Because you can’t rely on private-sector infrastructure to take on a massive public responsibility in disasters without proper planning and resources,” the authors say.
In what’s become a recurring theme during the pandemic, another NEJM perspective addresses shortages of critical supplies, particularly ventilators and personal protective equipment. “As the United States braces for a growing wave of patients with COVID-19 in our hospitals and ICUs, we must ensure that we have the key equipment needed to care for patients and to keep our healthcare workforce safe,” the authors argue.
The US Food and Drug Administration has taken additional steps to respond to the pandemic: issuing a temporary policy regarding the manufacture of alcohol for hand sanitizer products, adding hydroxychloroquine sulfate to category 1 (“bulk drug substances under evaluation”) under the interim policy on compounding using bulk drug substances, and issuing an emergency use authorization for ventilators.
In what will be a biweekly survey of members of the American Society of Health-System Pharmacists, 15% of respondents said the disruption of supplies of surgical-type masks was either major or moderate, “indicating either going without masks or using alternatives with mixed or poor results.” Another 39% reported minor disruptions that required use of acceptable alternatives.
The American College of Physicians “is calling for assessment of nonurgent, in-person medical appointments, and recommends transitioning patients who do not need to be seen in person to virtual visits, or delaying their visits during the national emergency created by COVID-19.”
Researchers report in JAMA on the feasibility of using elastometric half-mask respirators that are used in construction and manufacturing to address the shortfall of N95 respirators for healthcare workers.
In a JAMA viewpoint, Mary McDermott, MD (Northwestern University, Chicago, IL), and Anne Newman, MD (University of Pittsburgh, PA), discuss ways to preserve clinical trial integrity during the pandemic. “The effects of the coronavirus pandemic on randomized trials hopefully will be short-lived, relative to the long-term benefits of these trials to millions of people who will continue to live with debilitating medical diseases after the crisis ends,” they write. “To maximize public health benefits, creativity and persistence are required, especially during these unprecedented and uncertain times.”
The US Federal Emergency Management Agency (FEMA) has put up a “coronavirus rumor control” website.
In JAMA Network Open, Chinese researchers report on factors associated with mental health outcomes among healthcare workers exposed to COVID-19. A substantial number of workers—particularly women, nurses, those in Wuhan (where the virus is believed to have originated), and frontline workers—reported depression, anxiety, insomnia, and distress.
March 25, 2020
TCTMD’s Yael Maxwell explores how telehealth is emerging as a critical tool to connect physicians and other healthcare professionals with patients who are dealing with chronic cardiovascular conditions during the COVID-19 pandemic.
The European Society of Cardiology (ESC) has a couple of new videos on its COVID-19 page, one addressing questions about the clinical presentation of respiratory failure in COVID-19 and the other containing two important messages from ESC President Barbara Casadei, MD, DPhil (University of Oxford, England): don’t forget the needs of patients with CVD during the pandemic and also lobby with local/national governments for lockdowns to protect patients.
Scientists studying the SARS-CoV-2 virus say it’s not mutating quickly, suggesting that once a vaccine becomes available—which could take a year or more—it will offer long-lasting protection, according a news story in the Washington Post.
 In the context of continuing shortages of filtering facepiece respirators (FFRs) during the pandemic, the US Food and Drug Administration “has concluded based on the totality of scientific evidence available that certain imported disposable FFRs that are not NIOSH-approved are appropriate to protect the public health or safety.” Read the letter on the emergency use authorization here.
In the context of continuing shortages of filtering facepiece respirators (FFRs) during the pandemic, the US Food and Drug Administration “has concluded based on the totality of scientific evidence available that certain imported disposable FFRs that are not NIOSH-approved are appropriate to protect the public health or safety.” Read the letter on the emergency use authorization here.
The FDA has issued a warning to be on the lookout for fraudulent COVID-19 tests, vaccines, and treatments.
A viewpoint in JAMA tackles off-label drug use, compassionate use, and the conduct of randomized trials during pandemics. “The rapid and simultaneous combination of supportive care and randomized controlled trials is the only way to find effective and safe treatments for COVID-19 and any other future outbreak,” Andre Kalil, MD (University of Nebraska Medical Center, Omaha), says.
A JAMA Cardiology viewpoint by Chengzhi Yang, MD, PhD, and Zening Jin, MD, PhD (Beijing Tiantan Hospital, China), discusses the confluence of the acute respiratory infection of COVID-19 and cardiovascular disease.

Regulators from around the world meeting under the umbrella of the International Coalition of Medicines Regulatory Authorities (ICMRA) recently convened a workshop—co-chaired by the European Medicines Agency and the FDA—to lay out data requirements for phase I COVID-19 vaccine trials. The resulting report was released Tuesday.
March 24, 2020

Another major meeting has gone down to COVID-19. The Society for Cardiovascular Angiography and Interventions (SCAI) says that its annual scientific sessions scheduled to take place in Atlanta, GA, in the middle of May have been canceled, with plans being developed for virtual offerings. SCAI also announced that it is launching a COVID-19 resource center to help interventionalists navigate their way through the pandemic.
The American Heart Association (AHA) has put up $2.5 million to fast-track research focused on cardiovascular and cerebrovascular issues in the context of COVID-19. Grants will be given for projects that can provide results in 9 to 12 months. The AHA has also released interim guidance on reducing SARS-CoV-2 transmission during resuscitation care.
The Circulation COVID team has launched a series of short video interviews—called Updates from the Front Lines—with experts from pandemic hot spots around the world to gain insights into CV presentations, diagnostics, patient and practice management decisions, and more. The first video is with April Stempien-Otero, MD (University of Washington, Seattle).

Beware of promises of medicines that can prevent or treat COVID-19 or of easy access to legitimate medicines that might not be readily available during the pandemic from unregistered websites, the European Medicines Agency (EMA) says. In its warning, the EMA notes that there are currently no treatments approved for use against COVID-19.
Experts from Banner Health issued a warning of their own about self-medicating to prevent or treat COVID-19 after a man and his wife, both in their 60s, ingested chloroquine phosphate, “an additive commonly used at aquariums to clean fish tanks.” The man has died. Chloroquine, an antimalarial that has been proposed as a potential treatment of COVID-19 based on very preliminary evidence, should not be taken to prevent or treat the disease, the experts stressed.
A team led by Asif Qasim, MBBChir, PhD (King’s College Hospital, London, England), founder and CEO of MedShr, has launched a website—LetsBeatCOVID.net—to help hospitals and governments collect data from the public that can be useful while dealing with the outbreak. It contains an anonymous survey to collect data on people’s health and exposure to COVID-19.
March 23, 2020
In a video on Rutherford Medicine, interventional cardiologist Rasha Al-Lamee, MBBS (Imperial College London, England), describes how the emergence of COVID-19 has turned her day-to-day upside down: “This has been one of the most surreal weeks of my life, and I think that’s the same for clinicians all over the world at the moment.”
@rallamee joined us live via video link from Hammersmith Hospital.
— Rutherford Medicine (@rutherfordmed) March 23, 2020
She gave us a first-hand perspective of the challenges that cardiologists are facing every day during the COVID-19 pandemic.
Join the live discussion here:https://t.co/nWiMOgfXHA pic.twitter.com/Y8RgnDglVu
The novel coronavirus behind COVID-19 (SARS-CoV-2) has spread so quickly due to undocumented infections, a study published last week in Science suggests. Models based on reported infections in China indicates that 86% of all infections were undocumented before travel restrictions implemented on January 23. Undocumented infections were the source of 79% of documented cases, the authors report. “These findings explain the rapid geographic spread of SARS-CoV-2 and indicate containment of this virus will be particularly challenging,” they say.
Responding to reports on social media and elsewhere, the European Medicines Agency (EMA) says there is no evidence showing that ibuprofen is associated with worsening of COVID-19, adding that it “is monitoring the situation closely and will review any new information that becomes available on this issue in the context of the pandemic.” There remains a need for epidemiological studies on the effects of any of the NSAIDs on COVID-19, the agency says. The US Food and Drug Administration has also weighed in.
Regulators have recognized that the pandemic will impact ongoing clinical trials. Last week, the FDA released guidance on the management of trials in the context of the COVID-19 situation. The EMA, along with the European Commission and the Head of Medicines Agency, followed a few days later with advice of their own.
 The FDA has responded to COVID-19 in other ways, too. On Friday, the agency announced a new policy that allows for the expanded use of certain FDA-cleared, noninvasive devices for measuring vital signs so clinicians can monitor their patients remotely. And on Sunday, it issued guidance around risk evaluation and mitigation strategy (REMS)-required laboratory testing and imaging studies, saying clinicians should “consider whether there are compelling reasons not to complete these tests or studies during this public health emergency and use their best medical judgment in weighing the benefits and risks of continuing treatment in the absence of laboratory testing and imaging studies.”
The FDA has responded to COVID-19 in other ways, too. On Friday, the agency announced a new policy that allows for the expanded use of certain FDA-cleared, noninvasive devices for measuring vital signs so clinicians can monitor their patients remotely. And on Sunday, it issued guidance around risk evaluation and mitigation strategy (REMS)-required laboratory testing and imaging studies, saying clinicians should “consider whether there are compelling reasons not to complete these tests or studies during this public health emergency and use their best medical judgment in weighing the benefits and risks of continuing treatment in the absence of laboratory testing and imaging studies.”
Lopinavir-ritonavir does not look like a therapeutic solution for adults hospitalized with severe COVID-19. Published in the New England Journal of Medicine, a randomized trial of patients with laboratory-confirmed SARS-CoV-2 infection showed that those who received the treatment twice a day for 14 days on top of standard care did not have a shorter time to clinical improvement or a significantly lower mortality rate at 28 days.
Two NEJM articles published Monday tackle the difficult choices healthcare workers are forced to make when supplies are limited. Ezekiel Emanuel, MD, PhD (University of Pennsylvania, Philadelphia), et al discuss how to fairly distribute scarce medical resources, while Robert Truog, MD (Harvard Medical School and Boston Children’s Hospital, MA), and colleagues zero in on allocation of ventilators when there are not enough to go around.
COVID-19: TCTMD's Daily Dispatch for September
COVID-19: TCTMD’s Daily Dispatch for August
COVID-19: TCTMD’s Daily Dispatch for July
COVID-19: TCTMD’s Daily Dispatch for June
COVID-19: TCTMD’s Daily Dispatch for May
COVID-19: TCTMD’s Daily Dispatch for April
Todd Neale is the Associate News Editor for TCTMD and a Senior Medical Journalist. He got his start in journalism at …
Read Full Bio

Comments